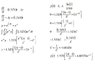- #1
6Stang7
- 212
- 0
Homework Statement
In an experiment, the isotope I-128 is created by bombarding I-127 with neutrons. This creates 1.5E6 I-128 atoms per second. Initially there are no I-128. The half-life of I-128 is 25 minutes. Find the equation that represents the number of I-128 atoms as a function of time.
Homework Equations
dy/dt=creation rate-decay rate
The Attempt at a Solution
I set up dy/dt=(1.5E6)t-ay.
a is the decay rate in e^(-at), and y(t) is the number of I-128 after time t.
I tried solving this as a first order linear equation and got a horriable answer. Where am I going wrong?