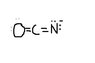- #1
icystrike
- 445
- 1
Homework Statement
Assign formal charges to the possible lewis structures of the following and decide which structure is the most important:
[tex]OCH^{-}[/tex]
Homework Equations
The Attempt at a Solution
Most important
I'm wondering how can i come up with all possiblities..