g.lemaitre
- 267
- 2
Here's a pic from Sean Carroll's lectures on Dark Matter and Dark Energy available through the Teaching Company:
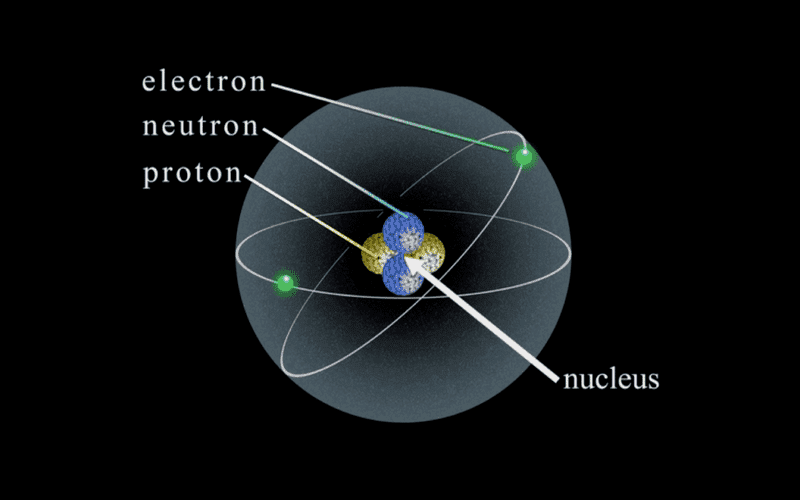
I just want to double check, there are two types of atoms, if I understand the delayed choice experiment correctly, just the knowledge that you can detect the atom transforms it from occupying a probability cloud (wave-like) where the electron does not really occupy a point in space to occupying a definite place (particle-like). I'm pretty sure I'm right and I don't understand why Carroll didn't throw in a few quantum mechanical slogans such as knowing that you can detect an atoms transforms it from x to y.
I just want to double check, there are two types of atoms, if I understand the delayed choice experiment correctly, just the knowledge that you can detect the atom transforms it from occupying a probability cloud (wave-like) where the electron does not really occupy a point in space to occupying a definite place (particle-like). I'm pretty sure I'm right and I don't understand why Carroll didn't throw in a few quantum mechanical slogans such as knowing that you can detect an atoms transforms it from x to y.