Discussion Overview
This thread discusses several chemistry-related homework questions from the 2012 GCE AL Sri Lanka exam, focusing on organic compounds, oxidation states, reactions, and acid strength. The scope includes problem-solving and conceptual understanding in organic chemistry.
Discussion Character
- Homework-related
- Debate/contested
Main Points Raised
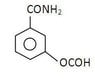
- Post 1 presents a series of questions and initial answers regarding organic compounds and reactions, including the oxidation state of sulfur in NSF and the reaction of an alkyne with NaHCO3.
- Post 2 agrees with most of Post 1's answers but challenges the assertion that the alkyne releases CO2 when reacting with NaHCO3, suggesting that alkynes are not strong enough acids for this reaction.
- Post 3 expresses gratitude for the feedback and acknowledges a mistake in the fifth question regarding the order of acid strength.
- Post 4 questions whether Post 3 corrected their understanding of the third question after receiving feedback.
- Post 5 reflects on the confusion regarding the CO2 release, indicating a reconsideration of their understanding.
Areas of Agreement / Disagreement
Participants generally agree on the correctness of most answers except for the reaction involving NaHCO3, where there is disagreement about whether CO2 is released. The discussion remains unresolved regarding the specifics of that reaction.
Contextual Notes
There are unresolved assumptions regarding the strength of alkynes as acids and the conditions under which the reactions occur. The discussion does not clarify the definitions of terms used or the context of the reactions.