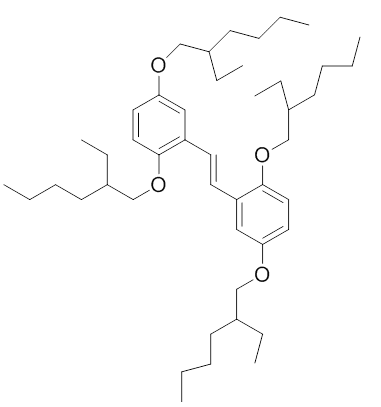
Discussion Overview
The discussion revolves around identifying which materials qualify as conjugated polymers, focusing on the definitions and characteristics that distinguish them from non-conjugated materials. Participants explore the structural requirements, such as the presence of pi bonds and the arrangement of single and double bonds, as well as the implications of aromaticity.
Discussion Character
- Exploratory
- Technical explanation
- Debate/contested
Main Points Raised
- One participant seeks clarification on which materials are conjugated polymers and expresses difficulty in identifying them.
- Another participant questions the definition of conjugated polymers and the role of pi bonds in their identification.
- It is suggested that the presence of alternating single and double bonds is necessary for a molecule to be considered conjugated.
- A participant challenges the assertion about alternating bonds in a specific molecule, indicating a disagreement on the structural interpretation.
- There is a discussion about whether round molecules can exhibit alternation in bonding and how that relates to conjugation.
- One participant posits that for a polymer to be considered conjugated, the conjugated bond system must extend throughout the entire molecule.
- Another participant expresses confusion about the concept of alternation in the context of aromatic compounds and their properties.
Areas of Agreement / Disagreement
Participants do not reach a consensus on the criteria for identifying conjugated polymers, with multiple competing views presented regarding the structural requirements and the role of aromaticity.
Contextual Notes
Limitations include varying interpretations of what constitutes alternating bonds, the impact of molecular shape on conjugation, and the specific requirements for aromatic compounds in relation to conjugation.
Who May Find This Useful
Individuals interested in polymer chemistry, materials science, and the structural characteristics of conjugated materials may find this discussion relevant.