Discussion Overview
The discussion centers around the adaptation of the chlor-alkali process to synthesize other salts, specifically focusing on the potential use of KNO3 to produce KOH and Cu(NO3)2. Participants explore the feasibility of using a salt bridge in an electrolytic cell setup and the implications of different electrode materials and configurations.
Discussion Character
- Exploratory
- Technical explanation
- Debate/contested
- Mathematical reasoning
Main Points Raised
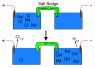
- One participant proposes using a salt bridge with KNO3 to produce KOH in one cell and Cu(NO3)2 in another, powered by a 9.6V battery.
- Another participant challenges the understanding of the chlor-alkali process and the proposed setup, suggesting a lack of clarity in the chemistry involved.
- A participant acknowledges a mistake in their initial description of the cell setup and attempts to clarify their understanding of the chlor-alkali process.
- There is a discussion about the movement of copper ions and the potential need to use only carbon electrodes to avoid complications with copper migration.
- Some participants express that while the proposed method may work to some extent, the electricity costs might outweigh the benefits of synthesizing KOH compared to purchasing it directly.
- One participant suggests that the ultimate goal is to isolate the nitrate ion for further synthesis rather than producing KOH.
- Another participant shares an idea about using solar cells to power the process, discussing the potential for refining copper and the challenges posed by other elements in the solution.
Areas of Agreement / Disagreement
Participants express differing views on the feasibility and understanding of the proposed adaptations to the chlor-alkali process. There is no consensus on whether the proposed cell setup will function as expected, and multiple competing views regarding the chemistry and practicality remain evident.
Contextual Notes
Participants highlight various assumptions about the behavior of ions in the electrolytic cell, the role of different electrode materials, and the implications of energy costs. There are unresolved questions regarding the efficiency and practicality of the proposed methods.
Who May Find This Useful
This discussion may be of interest to those exploring electrochemical processes, particularly in the context of synthesizing salts and refining metals, as well as individuals considering alternative energy sources for chemical processes.