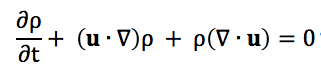So, not to be overly contrarian here, but I don't entirely agree with the analysis of
@bigfooted. While I do recognize that flow with low velocities and density variations do exist, I will contend that these are still
compressible flows even though they obey the Mach number requirement for incompressible flows. At the bottom of this post are a list of references I have laying around that all cite ##\rho=\mathrm{constant}## as the defining characteristic of an incompressible flow. The references span a range of levels (undergraduate, graduate, etc.).
The reason I favor such a definition is that the incompressible assumption commonly carries with it a handful of implications. First, if you assume the density to be constant, you are left with 4 equations and 4 unknowns and it decouples the energy equation from the system, so there is no closure problem. Second, while it is not enforced, the assumption of constant density carries with it the assumption that temperature variations are either nonexistent or quite small (otherwise, the constant density assumption doesn't make a lot of sense in reality), so that also let's us make a number of simplifying assumptions on the momentum and energy equations, such as that the viscosity and the thermal conductivity are constants, which let's us keep our relatively tractable 4 equations.
Generally speaking, if you want to capture all of the physics of flows that have variable density, you simply have to use the full compressible Navier-Stokes equations and treat it as a compressible flow, even if the flow itself is low speed. However, it is fairly common to
approximate such flows as incompressible to make the problem more tractable. Usually, in my experience, the approach in these situations is to assume the density variations are small and invoke the Boussinesq approximation. Usually that takes the form of assuming that, for reference (mean) values of ##\rho##, ##T##, and ##p##, the deviations are small and a Taylor Expansion yields
[tex]\rho = \rho_{\infty} + \left(\dfrac{\partial \rho}{\partial T}\right)(T - T_{\infty}) + \left(\dfrac{\partial \rho}{\partial p}\right)(p - p_{\infty})+H.O.T..[/tex]
Based on the values of these derivatives, this is often written as
[tex]\rho \approx \rho_{\infty} + \rho_{\infty}\beta(T - T_{\infty}) + \dfrac{\gamma}{c^2}(p - p_{\infty})[/tex]
where ##\beta## is the coefficient of thermal expansion, ##\gamma## is the ration of specific heats, and ##c## is the speed of sound. As long as the speed is low and the pressure variations are therefore fairly small, that ##c^{-2}## term is generally very small, so in this limit, the density's dependence on pressure is effectively negligble. In that sense, you can transform the full compressible equations into a simpler set that retain many of the features of the incompressible equations, but it's still not truly an incompressible flow. For larger variations in density, other methods are necessary to avoid using the compressible equations.
The one text that I have that doesn't define incompressibility is http://amzn.com/0521663962 . He uses the following definition: "A fluid is said to be
incompressible when the density of an element of fluid is not affected by changes in pressure." This is equivalent to saying that
[tex]\dfrac{D\rho}{Dt}=0[/tex]
or
[tex]\nabla\cdot\vec{V} = 0,[/tex]
that the velocity field is solenoidal. This is the same as what
@bigfooted has done. However, in the same text, the author goes on to use scaling analysis to describe the situations in which this assumption holds, and it boils down to five different criteria.
First:
[tex]\dfrac{V^2}{c^2} \ll 1[/tex]
This is the common criteria that the Mach number must be small, usually taken to be ##M<0.3## such that the above is more than an order of magnitude less than 1. This is violated in high speed aerodynamics and is probably the most commonly studied situation in which flows are compressible. This corresponds with the field of gas dynamics.
Second:
[tex]\dfrac{f^2 L^2}{c^2} \ll 1[/tex]
This pertains to unsteady flows where ##f## is some dominant frequency involved with the unsteadiness, ##L## is the characteristic length of a flow being considered, and ##c## is as above. This is effectively a squared Strouhal number based on sound speed. However, it approaches unity when considering sound waves, where ##L## corresponds to the wavelength of an unsteady motion of speed ##c## and frequency ##f##. In that case, the compressibility of the fluid (even liquids like water) is inherently important. This corresponds with the field of acoustics.
Third:
[tex]\dfrac{gL}c^2 \ll 1[/tex]
This is a statement about body forces balanced by pressure distributions. He shows that this really can only ever be violated in gases like air, and that it boils down to a criteria for whether the scale of the flow is small compared to ##p/(\rho g)##, which is called the scale height of the atmosphere and is something like 8+ km for air in our atmosphere. This criteria is violated in meteorology.
Fourth:
[tex]\dfrac{\beta U^2}{c_p}\dfrac{\mu}{\rho L U} \ll 1[/tex]
where ##\beta## is the coefficient of thermal expansion, ##\mu## is the viscosity, and ##c_p## is the specific heat at constant pressure. This relates to viscous dissipation being small enough that it does not generate enough heat to affect the density. Given the nature of viscous dissipation, it is almost impossible for this to be a factor, and I don't know of any situations off the top of my head in which this would absolutely be violated.
Fifth:
[tex]\beta\theta\dfrac{\kappa}{LU} \ll 1[/tex]
where ##\kappa## is the thermal diffusivity. This is a statement about heat conduction being small and not affecting the temperature enough to change the density. The only way this is violated is when you have a very high temperature gradient and/or small values of ##LU##. The candle situation actually typically can violate this. There is an analogous term for flow in which there are concentration or species variations to cover mass transfer and its effect on compressibility as well.
So, with all of that in mind, I would argue that the candle situation is
not incompressible in the strict sense of the term, and that ##\rho## being constant essentially covers all of the bases provided by Batchelor and is the better, more intuitive description for when a flow is incompressible.
Sources using ##\rho=\mathrm{constant}##:
http://amzn.com/1118912659
http://amzn.com/1118116135
http://amzn.com/1259129918
http://amzn.com/0073398276
http://amzn.com/0072402318
http://amzn.com/3540662707