r12214001
- 24
- 2
- Homework Statement
- HF molecular orbital
- Relevant Equations
- HF molecular orbital
In figure. Why F use 2P rather than 2S to bonding in HF MO model.
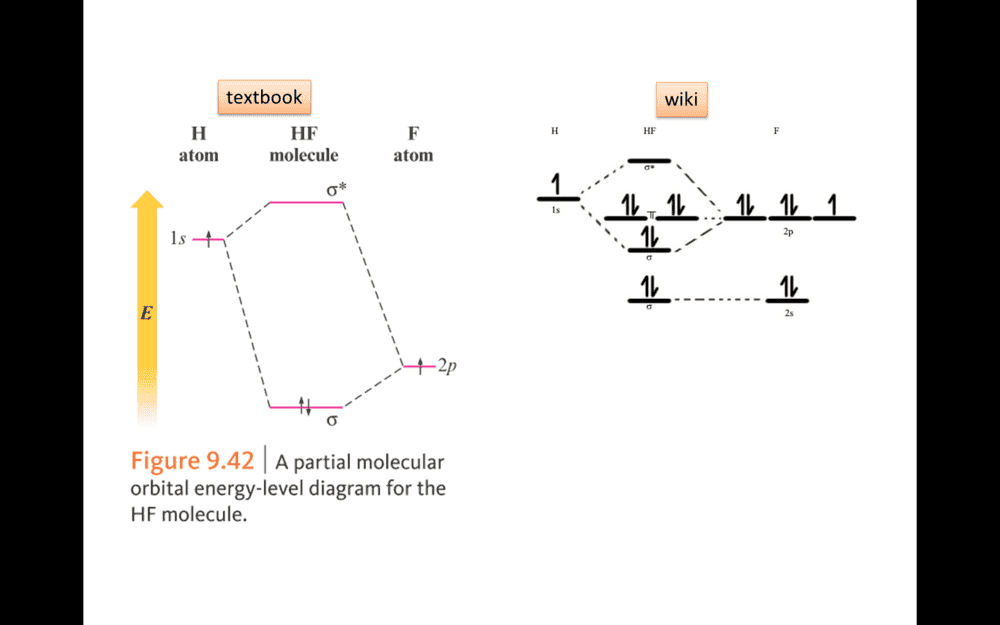
The discussion revolves around the bonding characteristics of fluorine in the context of the molecular orbital (MO) model for HF (hydrogen fluoride). Participants explore the use of different atomic orbitals in bonding, specifically questioning why fluorine utilizes 2P orbitals instead of 2S orbitals in this model.
The discussion contains multiple competing views regarding the role of hybridization and the specific orbitals involved in bonding for fluorine, indicating that there is no consensus on these points.
Participants express uncertainty about the applicability of hybridization in the context of molecular orbital theory and the specific orbital contributions to bonding in HF.
Readers interested in molecular orbital theory, atomic structure, and the bonding characteristics of elements, particularly in the context of fluorine and its compounds.
1S2 2S2 2P5Borek said:What is the electron configuration of a free F atom?
OK i get it. I should never use hybridization to account for MO theory.chemisttree said:Can you bond using suborbitals that are already filled?