Discussion Overview
The discussion revolves around calculating the total cooling effect in Watts of a system involving a heat pump and a cooling battery. Participants are sharing various data points related to the system's components, including temperatures, flow rates, and capacities, while seeking clarification on terms and system configuration.
Discussion Character
- Technical explanation
- Debate/contested
- Exploratory
Main Points Raised
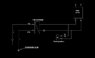
- One participant inquires about additional data needed to approximate the cooling effect, suggesting that specific flow rates and temperatures through the system are relevant.
- Another participant clarifies that "THR" stands for Thermal Heat Rejection and describes the cooling battery as a cooling element in an air handling unit, questioning the flow direction and configuration of the system.
- Participants discuss the flow rates and temperatures associated with the heat pump and cooling battery, with one providing specific values for the evaporator and cooling element.
- There is uncertainty regarding the configuration of the system, including the need for additional valves and the intended flow paths between components.
- One participant expresses that the term "THR" might be awkward if it refers to the heat removed from the water, indicating a lack of consensus on terminology.
Areas of Agreement / Disagreement
Participants have not reached a consensus on the configuration of the system or the implications of the data provided. Multiple competing views and uncertainties remain regarding the flow direction and the interpretation of terms used.
Contextual Notes
Limitations include unclear flow paths, potential missing assumptions about system configuration, and unresolved questions about the adequacy of the provided data for calculating the cooling effect.