Discussion Overview
The discussion revolves around the challenges faced in creating a cavity microelectrode for testing the electrochemistry of polyaniline powder. Participants share their experiences and suggestions regarding materials and methods for establishing electrical contact between the gold wire and the voltameter, as well as considerations for electrode design.
Discussion Character
- Exploratory
- Technical explanation
- Debate/contested
- Experimental/applied
Main Points Raised
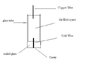
- One participant, Ryan, discusses the need for reliable contact between a gold wire and a voltameter, mentioning previous attempts with mercury that were unsuccessful due to gold dissolution.
- Ryan proposes three alternatives: using graphite powder, soldering the gold wire to a copper wire, or finding another low melting point metal.
- Another participant questions the necessity of soldering copper to gold, suggesting that a 4-point probe might be a better choice for the intended application.
- A different participant shares their positive results using a platinum wire with a mercury contact, noting the importance of avoiding redox potentials on the surface.
- This participant mentions their decision to use solder for contact, citing a melting point lower than that of the glass tube, and considers switching to platinum if gold proves ineffective.
- They also mention using an iron wire instead of copper for connection to the voltameter, emphasizing the need to avoid excessive use of platinum or gold.
- Another participant reflects on their past experience with liquid crystal conducting organic compounds and the 4-point probe, but indicates they are not currently up to date with the latest methods.
Areas of Agreement / Disagreement
Participants express various approaches and materials for achieving contact in cavity microelectrodes, with no clear consensus on the best method. Some suggest alternatives while others share their experiences, indicating a range of opinions and practices.
Contextual Notes
Participants reference different electrode configurations, such as 3-point and 4-point probes, without resolving which is definitively superior for the application at hand. There are also mentions of specific materials and their properties, but no agreement on the optimal choice.