Discussion Overview
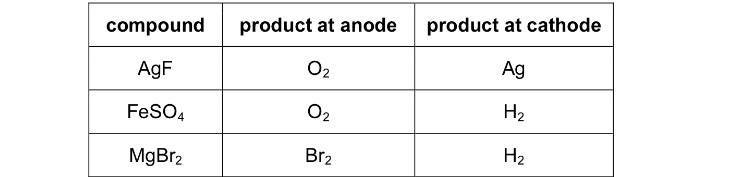
The discussion centers on the electrolysis of magnesium bromide (MgBr2) and the products formed at the anode, specifically questioning why bromine (Br2) is produced instead of oxygen (O2). The scope includes theoretical considerations of electrochemical reactions and kinetics involved in the process.
Discussion Character
- Homework-related
- Technical explanation
- Conceptual clarification
- Debate/contested
Main Points Raised
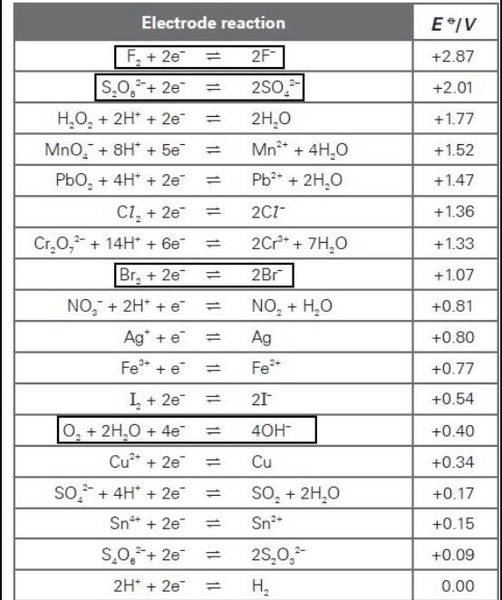
- One participant notes that the standard electrode potential (EΘ) values suggest that oxidation of hydroxide ions (OH-) is thermodynamically more favorable than that of bromide ions (Br-), which raises the question of why Br2 is produced.
- Another participant suggests that the kinetics of the reactions play a significant role, indicating that while oxygen should theoretically evolve, its production is slow and requires an overpotential, leading to the formation of Br2 at practical voltages.
- Several participants express uncertainty about the kinetics and the conditions under which different products may form, with one participant asking for clarification on other exceptions beyond brine and MgBr2.
- Repetitive assertions about the slow evolution of oxygen and the potential for other products to form at higher potentials are made, with participants advising caution regarding the reliability of these explanations.
Areas of Agreement / Disagreement
Participants generally agree that the kinetics of the reactions influence the products formed during electrolysis, but there is no consensus on the details of these processes or the implications for other scenarios.
Contextual Notes
There are unresolved aspects regarding the specific conditions that lead to the production of Br2 over O2, including the role of overpotential and the exact mechanisms at play in the electrolysis of MgBr2.
 [/B]
[/B]