Discussion Overview
The discussion revolves around the reaction of bromine (Br2) with certain organic compounds, focusing on identifying possible products and their stereoisomers. Participants explore the mechanisms of bromination, the formation of intermediates, and the implications for stereochemistry, particularly in the context of homework problems.
Discussion Character
- Homework-related
- Technical explanation
- Debate/contested
- Mathematical reasoning
Main Points Raised
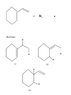
- Some participants propose that for product 1, there is 1 chiral center and 1 double bond, leading to a calculation of 22 products, while others suggest the correct answer is 2 products.
- There is confusion regarding the representation of products 1, 2, and 3, with some participants seeking clarification on whether they correspond to structures drawn by the original poster.
- Participants discuss the mechanism of bromine attachment, questioning whether it involves a planar intermediate or not, with some suggesting that the bromonium ion is non-planar.
- One participant notes that having a double bond does not guarantee stereoisomerism unless there is flexibility for multiple orientations, particularly in cyclic structures.
- There is a debate about whether cis and trans products are formed in the reaction, with some asserting that the bromide ion's attack leads to exclusively trans products.
- Some participants express uncertainty about how two stereocenters can be dependent on each other, with explanations provided regarding the orientation of bromine attachments.
- One participant concludes that only one product exists in trans form, while another argues that two products are possible due to the nature of stereocenter interactions.
Areas of Agreement / Disagreement
Participants do not reach a consensus on the number of products and stereoisomers resulting from the reaction. Multiple competing views exist regarding the mechanisms of bromination and the implications for stereochemistry.
Contextual Notes
Some participants highlight limitations in understanding the problem statement and the assumptions regarding the nature of the intermediates and the stereochemistry involved in the reaction.