Discussion Overview
The discussion revolves around the rate of stimulated emission in relation to spontaneous emission, particularly in the context of mathematical modeling as described in a Wikipedia article. Participants explore the conditions under which spontaneous emission can be neglected and the implications for the rate of change of population N2 in a system.
Discussion Character
- Technical explanation
- Debate/contested
Main Points Raised
- One participant expresses confusion about why the rate of change of N2 is attributed solely to stimulated emission, questioning the neglect of spontaneous emission.
- Another participant clarifies that the article focuses exclusively on stimulated emission, implying that spontaneous emission is not considered in that context.
- A further inquiry is made into the conditions under which spontaneous emission can be neglected, suggesting that it varies by system.
- Another participant elaborates that spontaneous emission is not neglected but rather not included by definition when calculating the rate of decay due to stimulated emission alone.
- The mathematical relationship involving the rates of spontaneous emission, stimulated emission, and absorption is presented, indicating that all three processes can be considered in a complete model.
- Equations relating the coefficients of these processes are discussed, including their dependence on thermodynamic equilibrium and Boltzmann statistics.
Areas of Agreement / Disagreement
Participants do not reach a consensus on the treatment of spontaneous emission, with some arguing it can be neglected in certain systems while others emphasize that it is not considered in the specific context of stimulated emission calculations.
Contextual Notes
The discussion highlights the complexity of modeling emission processes and the assumptions involved, particularly regarding the neglect of spontaneous emission in certain theoretical frameworks.
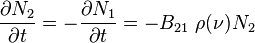 is true I would think that the rate in which N2 is changing is the rate of stimulated emission and spontanous emission together. Why is it just the rate of stimulated emission
is true I would think that the rate in which N2 is changing is the rate of stimulated emission and spontanous emission together. Why is it just the rate of stimulated emission