Discussion Overview
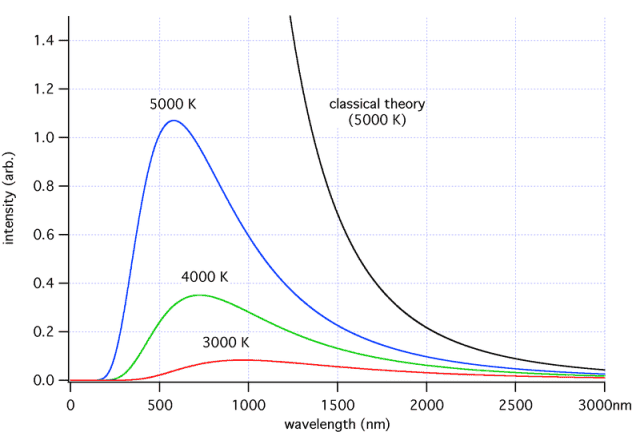
The discussion centers around the ultraviolet catastrophe, particularly in the context of black body radiation and the discrepancies between classical physics predictions and experimental observations. Participants explore the underlying theories, including the Rayleigh-Jeans law, and the implications of modes of oscillation in electromagnetic fields.
Discussion Character
- Exploratory
- Technical explanation
- Conceptual clarification
- Debate/contested
Main Points Raised
- Some participants seek to understand why classical theory predicts that intensity increases with frequency, referencing the Rayleigh-Jeans law.
- Others explain that classical theory counts the number of modes in each frequency interval, leading to the conclusion that there are infinitely many modes at high frequencies, which contributes to the ultraviolet catastrophe.
- A participant proposes that the increase in modes at higher frequencies may be due to a larger combination of the substance's degrees of freedom, suggesting that higher frequency radiation involves more complex interactions.
- Another participant describes the relationship between electromagnetic modes and their frequencies using a geometric analogy involving points in a spherical shell.
- Some participants discuss the concept of standing waves and how they relate to the modes of oscillation, questioning how traveling waves can be viewed as standing waves.
- One participant introduces the idea that the electromagnetic field can be analyzed through its Fourier components, linking the intensity of radiation to the amplitudes of these modes.
- There is a claim that intensity decreases at higher frequencies, which contradicts earlier points about classical predictions.
- A participant mentions the energy-momentum tensor for photons, discussing the relationship between energy and momentum in the context of phase space.
Areas of Agreement / Disagreement
Participants express a range of views on the nature of modes and their implications for black body radiation. There is no consensus on the interpretation of how modes contribute to intensity or the validity of classical predictions versus experimental results.
Contextual Notes
Some discussions involve unresolved assumptions about the nature of modes and their relationship to energy distribution, as well as the implications of the superposition principle in electromagnetic theory.