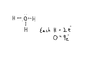Discussion Overview
The discussion revolves around the charge states of the hydronium ion (H3O+) and the isocyanate ion (NCO-), exploring the reasoning behind their respective charges. Participants engage in a technical examination of electron counting, the octet rule, and Lewis structures, with a focus on theoretical and conceptual aspects of chemistry.
Discussion Character
- Technical explanation
- Conceptual clarification
- Debate/contested
- Mathematical reasoning
Main Points Raised
- Some participants assert that H3O+ has an extra electron, questioning its positive charge, while others clarify that it does not have an extra electron and emphasize the importance of proper electron accounting.
- There is a contention regarding the number of electrons associated with oxygen in H3O+, with some claiming it has 9 electrons and others correcting that it has 6 valence electrons, needing 2 more to satisfy the octet rule.
- Participants discuss the concept of formal charge and how it applies to the hydronium ion, with references to diagrams and tutorials to support their claims.
- Some argue that the octet rule is not a strict rule but a guideline, suggesting that the understanding of electron configurations should not be limited to this concept.
- There is a proposal that the formation of H3O+ can be understood through the dissociation of water, leading to the combination of H+ and H2O.
- Several participants express uncertainty about the best way to conceptualize the charge states and the electron configurations involved in the formation of H3O+ and NCO-.
Areas of Agreement / Disagreement
Participants do not reach a consensus; there are multiple competing views regarding the electron configurations and charge states of H3O+ and NCO-. The discussion remains unresolved with ongoing debate about the interpretations of the octet rule and the implications for molecular stability.
Contextual Notes
Limitations include varying interpretations of the octet rule, differing assumptions about electron sharing in covalent bonds, and the complexity of Lewis structures. Some participants reference external resources for clarification, indicating a reliance on additional materials for understanding.
Who May Find This Useful
This discussion may be useful for students and educators in chemistry, particularly those interested in molecular structures, charge states, and the foundational concepts of electron configuration and bonding.