Discussion Overview
The discussion revolves around the search for a simple and convenient test to detect the presence of sugar or aspartame in beverages, particularly for individuals with diabetes. Participants explore various methods, including experimental approaches and theoretical considerations, without reaching a consensus on a definitive solution.
Discussion Character
- Exploratory
- Technical explanation
- Debate/contested
Main Points Raised
- One participant expresses a need for a cost-effective test to determine if their cola contains sugar instead of diet cola.
- Another participant shares results from using a blood glucose monitor on different cola samples, noting that the readings were inconsistent and often resulted in errors.
- Some participants suggest that taste differences between aspartame and sugar-sweetened colas could be a clue, but acknowledge this is not conclusive.
- A suggestion is made to use a brix refractometer, with links provided for purchasing or making one.
- Concerns are raised about whether aspartame has a different refractive index compared to sugar.
- One participant proposes a buoyancy test using cans of soda in water to differentiate between aspartame and sugar, although its application to fast food drinks is questioned.
- There are mentions of aspartame's instability under certain conditions, including its breakdown into methanol.
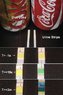
- Urine test strips are suggested as a potential solution, noted for their convenience and low cost.
- Some participants discuss the limitations of certain chemical tests, such as Benedict's test, in detecting specific types of sugars.
- One participant expresses a desire for a test that can be performed in a car.
Areas of Agreement / Disagreement
Participants do not reach a consensus on a specific method for testing sugar or aspartame in drinks. Multiple competing views and suggestions are presented, indicating ongoing uncertainty and exploration of the topic.
Contextual Notes
Some methods discussed may depend on specific conditions, such as the stability of aspartame or the type of sugar present, which could affect the reliability of the proposed tests.