Discussion Overview
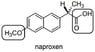
The discussion centers on determining the chirality of the molecule Naproxen using the Cahn–Ingold–Prelog (CIP) rules. Participants explore the identification of the stereocenter and the application of the CIP rules to assign priorities to the substituents attached to the chiral carbon.
Discussion Character
- Homework-related
- Technical explanation
- Debate/contested
Main Points Raised
- One participant identifies the stereocenter at the carbon where the carboxyl group meets the two benzene rings and expresses confusion about applying the CIP rules for determining priorities.
- Another participant confirms the identification of the chiral center and lists the three groups attached to it: a hydrogen, a carbon with multiple oxygen atoms, and a carbon with three carbon atoms.
- There is a discussion about how multiple bonds are treated in the CIP rules, with a participant recalling that multiple bonds count as equivalent single bonds.
- A participant questions how to determine priorities when substituents include aromatic rings, expressing uncertainty about which branch to follow in such cases.
Areas of Agreement / Disagreement
Participants generally agree on the identification of the chiral center, but there is uncertainty and confusion regarding the application of the CIP rules, particularly in cases with multiple bonds and complex substituents. The discussion remains unresolved regarding the correct prioritization in certain scenarios.
Contextual Notes
Participants express limitations in understanding the CIP rules, particularly in the context of multiple bonds and branching in substituents. There are unresolved questions about how to navigate these complexities when determining chirality.
Who May Find This Useful
Students studying organic chemistry, particularly those learning about chirality and stereochemistry, may find this discussion relevant.