Discussion Overview
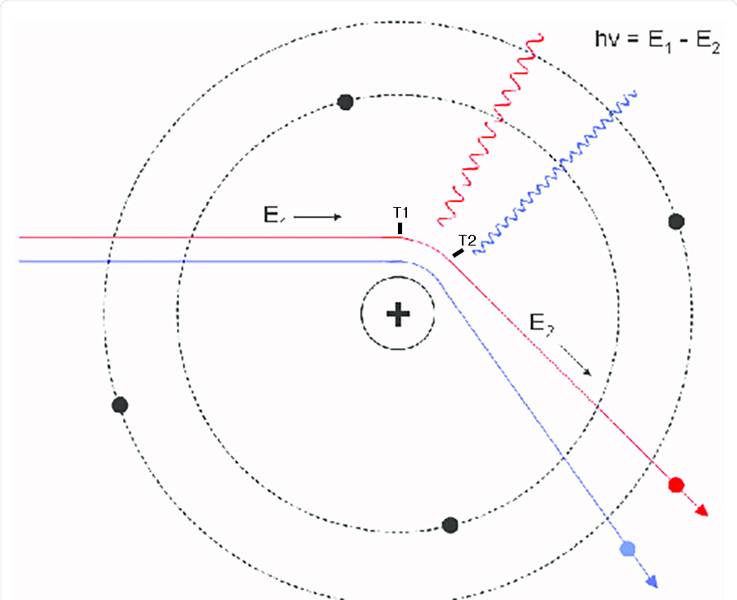
The discussion revolves around the implications of electron scattering off an atomic nucleus, particularly focusing on the relationship between the emitted bremsstrahlung radiation and the electron's acceleration and momentum changes. Participants explore concepts related to the timing of these processes, the nature of photon emission, and the rules governing transitions between quantum states.
Discussion Character
- Exploratory
- Debate/contested
- Technical explanation
- Conceptual clarification
Main Points Raised
- Some participants question whether the wavelength of emitted bremsstrahlung radiation can provide information about the duration of electron acceleration.
- Others argue that since the electromagnetic force is infinite in range, the electron is always accelerating, complicating the definition of a time interval for acceleration.
- There is a proposal to define a time period during which the electron's momentum vector changes significantly, and to correlate this with the wavelength of emitted photons.
- Some participants discuss the implications of selection rules for orbital transitions and whether these rules apply to unbound orbits, raising questions about the energies of emitted radiation.
- Concerns are raised about the instantaneous nature of photon emission and whether it can coexist with continuous changes in the electron's momentum vector.
- Participants express confusion regarding the relationship between the timing of photon emission and the electron's momentum changes, particularly in the context of energy conservation equations.
- There are discussions about the validity of classical treatments of electron behavior and the implications for photon absorption and emission processes.
Areas of Agreement / Disagreement
Participants do not reach consensus on the nature of the timing of electron acceleration and photon emission. Multiple competing views remain regarding the definitions of time intervals and the applicability of classical versus quantum descriptions.
Contextual Notes
Limitations include the ambiguity in defining time intervals for acceleration and emission processes, as well as the dependence on classical versus quantum mechanical interpretations. The discussion also highlights unresolved questions about the implications of selection rules and the instantaneous nature of photon interactions.