Discussion Overview
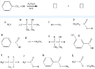
The discussion revolves around identifying the products of a chemical reaction involving potassium dichromate and an alcohol, with participants selecting from a list of compounds labeled A - L. The focus is on the oxidation process and the resulting products.
Discussion Character
- Exploratory
- Technical explanation
- Debate/contested
Main Points Raised
- One participant initially proposes that the product of the reaction is only K, but expresses uncertainty about this conclusion.
- Another participant questions the reasoning behind selecting K, specifically asking about the action of potassium dichromate on the alcohol.
- A hint is provided regarding the reputation of potassium dichromate, suggesting its role as an oxidizing agent.
- A later reply explains that potassium dichromate, in conjunction with hydrochloric acid, oxidizes the alcohol to produce a carboxylic acid, identifying the product as the compound represented by the letter E.
Areas of Agreement / Disagreement
Participants do not appear to reach a consensus, as there are differing views on the correct product of the reaction, with at least two competing claims regarding the products (K and E).
Contextual Notes
There are unresolved assumptions regarding the specific conditions of the reaction and the definitions of the compounds listed A - L. The discussion does not clarify the exact nature of the alcohol or the reaction conditions.