Discussion Overview
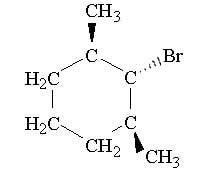
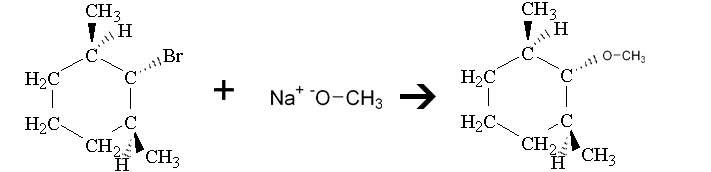
The discussion centers on the formation of substitution products from 1-bromo-2,6-dimethylcyclohexane when treated with sodium methoxide. Participants explore the mechanisms involved, specifically the potential for substitution versus elimination reactions, and the implications of the compound's structure on these processes.
Discussion Character
- Homework-related
- Technical explanation
- Debate/contested
Main Points Raised
- One participant suggests that sodium methoxide, being a weak base and a good nucleophile, favors an SN2 reaction, leading to a substitution product.
- Another participant asserts that sodium methoxide is a strong base that favors SN2 reactions, and corrects the product drawn by the original poster, emphasizing the rear-end attack of the nucleophile.
- A later reply questions the characterization of sodium methoxide as a strong base, stating it is just stable.
- Another participant explains that sodium methoxide is stable as a salt and, when in a polar solvent, dissociates into sodium cation and methoxide anion, with alkoxides being fairly strong bases.
- One participant hints that the requirement for elimination reactions involves the availability of hydrogen for abstraction and the energy of the transition state, suggesting that these factors may not favor elimination in this case.
- A suggestion is made to draw the most stable chair conformer of the reactant to understand why elimination may not occur.
- Participants are reminded to consider the stereochemistry changes that occur during SN1 or SN2 mechanisms when discussing the substitution products.
Areas of Agreement / Disagreement
Participants express differing views on the nature of sodium methoxide as a base and its implications for reaction mechanisms. There is no consensus on whether sodium methoxide should be classified as a strong base or not, and the discussion on elimination versus substitution remains unresolved.
Contextual Notes
There are unresolved assumptions regarding the energy requirements for elimination reactions and the specific structural characteristics of the compound that influence the reaction pathway. The discussion also reflects varying interpretations of the stability and reactivity of sodium methoxide.