mycotheology
- 86
- 0
I posted this in two forums because the question might be too chemistry related for the people in the calculus forum.
I'm a visual thinker so I struggle a bit to get my head around calculus concepts. So as an example, here's a potential energy surface:
lets say this represents the structure of a simple molecule like n-propane:
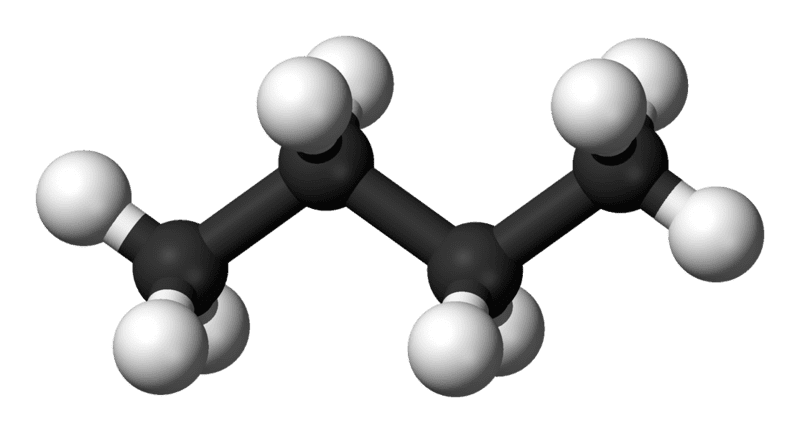
the molecule in the picture is the most stable conformation so the global minimum (the big pit) on the PES represents that conformation. Then the local minimum (the little bit) represents the staggered conformation. If I rotate or stretch the bonds in any other way, then it becomes less stable so the molecules energy will no longer be represented by a minimum on the PES.
I can see how the first derivative will help you locate the minima because the slope will be 0 there. The first derivative of this potential energy surface is called the "force" which makes sense to me because the y-axis represents the potential energy so when the molecule is in a stable conformation, the force which would ordinarily push the molecule into a stable conformation (if it was in an unstable one) is 0. What I'm trying to get my head around now is what the 2nd derivative represents. They call it the "force constant" and I know that in computational chemistry, they'll calculate the "force constant matrix" for a molecule. I'm trying to figure out what that means.
I'm a visual thinker so I struggle a bit to get my head around calculus concepts. So as an example, here's a potential energy surface:
lets say this represents the structure of a simple molecule like n-propane:
the molecule in the picture is the most stable conformation so the global minimum (the big pit) on the PES represents that conformation. Then the local minimum (the little bit) represents the staggered conformation. If I rotate or stretch the bonds in any other way, then it becomes less stable so the molecules energy will no longer be represented by a minimum on the PES.
I can see how the first derivative will help you locate the minima because the slope will be 0 there. The first derivative of this potential energy surface is called the "force" which makes sense to me because the y-axis represents the potential energy so when the molecule is in a stable conformation, the force which would ordinarily push the molecule into a stable conformation (if it was in an unstable one) is 0. What I'm trying to get my head around now is what the 2nd derivative represents. They call it the "force constant" and I know that in computational chemistry, they'll calculate the "force constant matrix" for a molecule. I'm trying to figure out what that means.
Last edited by a moderator: