Discussion Overview
The discussion centers around the comparison between graphyne and graphene, focusing on their mechanical properties, electron mobility, and potential applications. Participants explore theoretical aspects, synthesis challenges, and the implications of structural differences between the two materials.
Discussion Character
- Exploratory
- Technical explanation
- Debate/contested
Main Points Raised
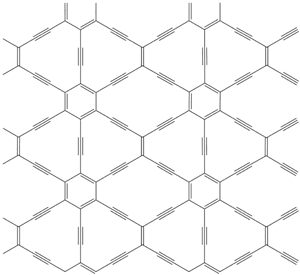
- Some participants suggest that graphyne's carbon-carbon triple bonds may weaken its mechanical properties due to a polar character affecting SP2-hybridization, while others propose that this polar character could enhance electron mobility and bandgap properties.
- There is speculation that graphyne may be lighter than graphene but could exhibit directional dependence in its mechanical properties, potentially making it less suitable for applications requiring uniform strength, such as super-strong cables.
- Some contributions indicate that graphyne might be harder to synthesize than graphene, raising questions about its practical applications.
- Participants reference studies indicating that graphyne's mechanical properties, including fracture strain and stress, depend significantly on the direction of applied strain, which contrasts with graphene's more isotropic behavior.
- There are claims that graphyne possesses unique electronic properties, including Dirac cones, which may differ from those of graphene due to its lack of hexagonal symmetry.
- A participant inquires about the synthesis methods for graphyne, indicating interest in the practical aspects of producing this material.
Areas of Agreement / Disagreement
Participants express a range of views on the mechanical and electronic properties of graphyne compared to graphene, with no consensus reached on which material is superior or the implications of their differences.
Contextual Notes
Some discussions involve assumptions about the mechanical and electronic properties based on structural characteristics, and there are references to ongoing research that may not fully resolve the comparisons being made.
Who May Find This Useful
This discussion may be of interest to researchers and students in materials science, nanotechnology, and those exploring advanced carbon allotropes and their applications.