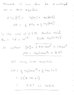Discussion Overview
The discussion revolves around the application of the Henderson-Hasselbalch (H-H) equation in calculating pH during the titration of a weak acid with a strong base, particularly at the equivalence point. Participants explore the theoretical underpinnings of the H-H equation, its assumptions, and the implications for pH calculations in different scenarios.
Discussion Character
- Debate/contested
- Technical explanation
- Mathematical reasoning
Main Points Raised
- Some participants assert that the H-H equation cannot be used at the equivalence point due to the significant presence of hydroxide ions ([OH-]) from the strong base, which violates the assumptions of the equation.
- Others argue that the H-H equation is valid as long as equilibrium concentrations are used, suggesting that it can be applied under certain conditions.
- One participant challenges the derivation of the H-H equation, questioning the assumptions made regarding the concentrations of the acid and its conjugate base.
- Another participant provides a derivation of the H-H equation and discusses its application to calculate the initial pH of a weak acid solution, emphasizing the importance of the [OH-] concentration.
- There are claims that the initial pH of a weak acid cannot be calculated using the H-H equation, similar to the situation at the equivalence point.
- Participants discuss the need for clarity in the derivation of equations and the conditions under which they apply, particularly regarding the assumptions about dissociation percentages.
Areas of Agreement / Disagreement
Participants generally disagree on the applicability of the H-H equation for calculating pH at both the equivalence point and the initial state of a weak acid solution. Multiple competing views remain regarding the assumptions and derivations related to the equation.
Contextual Notes
Limitations in the discussion include unresolved assumptions about the concentrations of species involved, the applicability of the H-H equation under varying conditions, and the mathematical steps required for accurate pH calculations.