Discussion Overview
The discussion revolves around the application of the oxidation number method in redox reactions, specifically focusing on a scenario where the same molecule, iodine, undergoes both oxidation and reduction. Participants are exploring how to determine the changes in oxidation numbers and how to proceed with calculations based on these changes.
Discussion Character
- Homework-related
- Technical explanation
Main Points Raised
- One participant notes that in the given problem, iodine is both oxidized and reduced, leading to two different changes in oxidation numbers (7 and 1).
- Another participant confirms the existence of these two different changes in oxidation numbers.
- A subsequent participant asks how to proceed after identifying the changes in oxidation numbers.
- One participant suggests that the approach is similar to other redox reactions, indicating that one half-reaction is for reduction and the other for oxidation, and they propose combining them.
- Another participant attempts to clarify the calculation process, suggesting a multiplication of coefficients to arrive at a final value.
- One participant expresses confusion about the explanation provided and requests further clarification.
- A later reply indicates that the participant has gained understanding after the discussion.
Areas of Agreement / Disagreement
Participants appear to agree on the method of using half-reactions for oxidation and reduction, but there is some confusion regarding the specific calculations and explanations. The discussion remains somewhat unresolved as participants seek clarity on the steps involved.
Contextual Notes
There are limitations in the clarity of the problem statement and the assumptions made about the oxidation states. The discussion does not fully resolve the mathematical steps involved in the calculations.
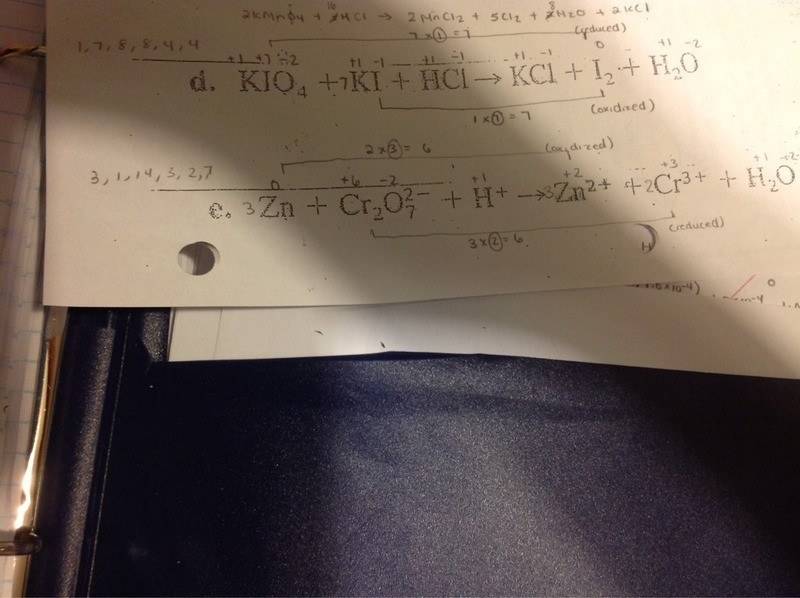 2) Relevant Equations3) Attempt at Solution
2) Relevant Equations3) Attempt at Solution