Discussion Overview
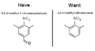
The discussion revolves around the conversion of 3,5-dimethyl-4-nitro-benzaldehyde to 2,6-dimethyl-nitrobenzene, specifically focusing on the removal of the aldehyde moiety. Participants explore various chemical methods, including oxidation and the Cannizzaro reaction, while addressing concerns about preserving other functional groups in the molecule.
Discussion Character
- Exploratory
- Technical explanation
- Debate/contested
- Mathematical reasoning
Main Points Raised
- Jeff expresses a desire to remove the aldehyde group without affecting the methyl groups, indicating a lack of experience in organic chemistry.
- Some participants suggest that mild oxidation might not affect the methyl groups, proposing reagents like PCC or silver ammonium nitrate.
- There is a discussion about the correct nomenclature for the compound in question, with Jeff acknowledging a mistake and seeking clarification.
- One participant proposes the Cannizzaro reaction followed by decarboxylation as a potential method for achieving the desired conversion.
- Concerns are raised about the possibility of concentrated base in the Cannizzaro reaction affecting the methyl groups, with some participants expressing doubt about this outcome.
- Another participant clarifies the advantages and disadvantages of the Cannizzaro reaction, emphasizing its oxidizer-free nature but noting potential yield issues.
- There is a reiteration of the Cannizzaro reaction process, with a focus on producing an alcohol and a carboxylic acid, and the subsequent steps to remove the acid while preserving the methyl groups.
Areas of Agreement / Disagreement
Participants do not reach a consensus on the best method for removing the aldehyde group, with multiple competing views on the effectiveness and safety of suggested reactions, particularly regarding the Cannizzaro reaction and oxidation methods.
Contextual Notes
Participants express uncertainty about the nomenclature and the specific reagents involved, indicating a reliance on various chemical reactions that may have different outcomes based on conditions and assumptions.
Who May Find This Useful
Individuals interested in organic chemistry, particularly those exploring functional group transformations and reaction mechanisms, may find this discussion relevant.