Discussion Overview
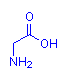
The discussion focuses on the basicity of different functional groups in glycine, specifically comparing the hydroxyl (OH) end and the amino (NH2) end to determine which is more likely to be protonated. The scope includes conceptual reasoning about molecular properties and comparisons with other similar molecules.
Discussion Character
- Conceptual clarification
- Debate/contested
Main Points Raised
- Some participants inquire about how to determine the basicity of the OH and NH2 ends of glycine without relying on memorized pKa values.
- One participant suggests making analogies with other molecules, such as CH3-OH versus CH3-NH2, to explore which is more protonable.
- Another participant points out that in amino acids, the carboxyl group is typically acidic (loses H+) while the amino group is basic (accepts H+), implying a straightforward way to identify acidic and basic ends.
Areas of Agreement / Disagreement
Participants express differing views on how to assess basicity, with no consensus reached on which end of glycine is more basic or the criteria for determining basicity.
Contextual Notes
Some discussions rely on analogies and comparisons, which may not fully account for the specific chemical environment of glycine. There is an implicit assumption that basicity can be determined through reasoning rather than empirical data.