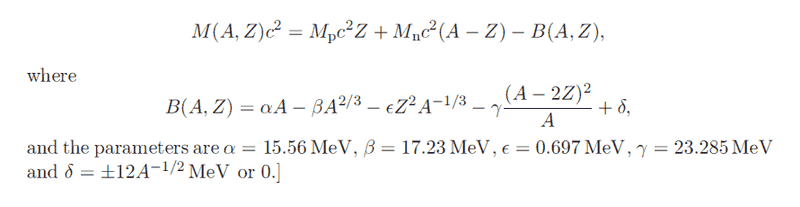Discussion Overview
The discussion centers around the beta decay of Rubidium-87 (##^{87}_{37}Rb##) to Strontium-87 (##^{87}_{38}Sr##), exploring the stability of Rubidium and the implications of its long half-life compared to other isotopes like Uranium-235 and Vanadium-50. The scope includes theoretical considerations of nuclear stability and decay rates.
Discussion Character
- Exploratory
- Technical explanation
- Debate/contested
Main Points Raised
- One participant calculates the most stable atomic number (Z) for a given mass number (A) and finds that for A=87, Z=38 is the most stable, questioning why Rubidium does not beta decay to Strontium.
- Another participant asserts that Rubidium does beta decay with a half-life of approximately 4.8 x 1010 years, referencing external sources.
- A participant questions the stability of Rubidium given its long half-life, prompting further inquiry into what the expected half-life should be.
- Another participant compares Rubidium's half-life to that of Uranium-235, suggesting that Rubidium should decay quicker if it is more stable to undergo beta decay.
- One participant mentions Vanadium-50's significantly longer half-life, contributing to the discussion about the variability of half-lives among different isotopes.
Areas of Agreement / Disagreement
Participants express differing views on the implications of Rubidium's half-life and its stability, with no consensus reached regarding the expected decay rates or the reasons behind the observed stability.
Contextual Notes
Participants do not clarify the assumptions behind their calculations or the definitions of stability and decay rates, leaving some aspects of the discussion unresolved.