Discussion Overview
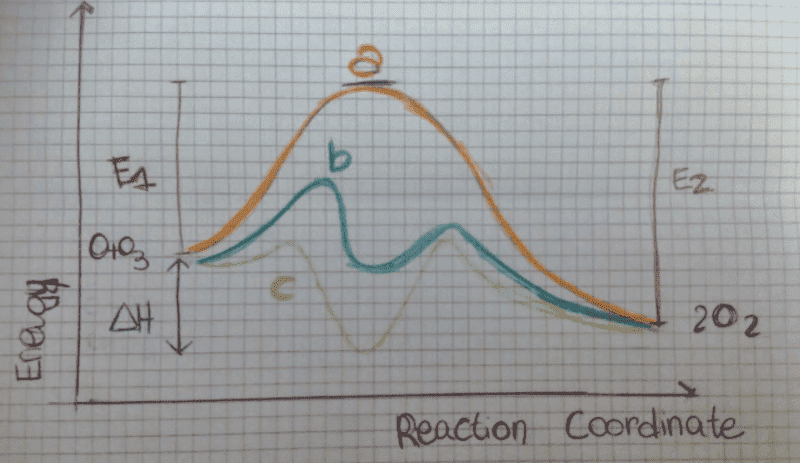
The discussion revolves around the effectiveness of two catalysts, 'B' and 'C', in a chemical reaction. Participants explore the reasons why catalyst 'C' is perceived as less effective than catalyst 'B', focusing on the concepts of activation energy, intermediate states, and the thermodynamic and kinetic aspects of the reaction.
Discussion Character
- Debate/contested, Technical explanation, Conceptual clarification
Main Points Raised
- Some participants suggest that catalyst 'C' is worse because, despite having a lower activation energy, the energy required for its intermediate state to evolve into products is higher than that for catalyst 'B'.
- Others argue that the intermediate state of catalyst 'C' is thermodynamically similar to the final products, which may lead to a higher likelihood of reverting to the initial state rather than proceeding to the final state.
- A participant mentions that both catalysts induce a change in mechanism and that while 'C' may create intermediates faster, it does not necessarily lead to a higher overall product accumulation compared to 'B'.
- There is a discussion about the probability of the intermediate state returning to the initial state versus progressing to the final state, with some participants noting that this probability is higher for catalyst 'C'.
- One participant acknowledges a misunderstanding regarding the energy potential barriers, indicating that the kinetic probabilities may not favor the final product as initially thought.
- Another participant asserts that the intermediate in catalyst 'C' is favored over the final product, supporting the view that catalyst 'B' is more effective in producing the final product.
Areas of Agreement / Disagreement
Participants express differing views on the effectiveness of catalyst 'C' compared to 'B', with no consensus reached. Some believe 'C' is less effective due to its intermediate state dynamics, while others argue that both catalysts have their merits in terms of reaction mechanisms.
Contextual Notes
Participants discuss the implications of thermodynamic and kinetic factors without resolving the complexities of the energy barriers and reaction pathways involved.