flintstones
- 21
- 1
- Homework Statement
- Explain why the alpha particle's path has a larger radius than either of the beta particle paths. Justify your answer using either momentum or charge-to-mass ratio.
- Relevant Equations
- Fc = Fm, so:
mv/r = qB
Suppose you are analyzing this image. The question to answer is: Explain why the alpha particle's path has a larger radius than either of the beta particle paths. Justify your answer using either momentum or charge-to-mass ratio.
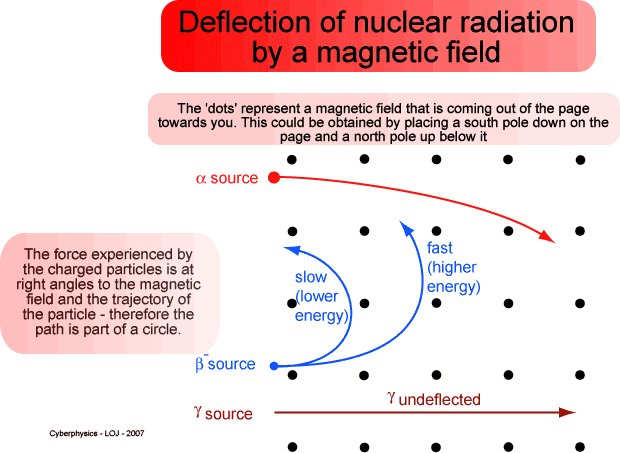
Momentum
p = qBr
As long as q and B are constant, the greater r is, the greater p will be. However, q is not constant here, since alphas carry double the charge magnitude of betas. So I'm not sure you can confidently say that momentum is the reason the alpha and beta have different radii. Am I wrong here?
Charge to Mass Ratio
q/m = v/(Br)
As long as v and B are constant, the greater r is, the smaller the charge-to-mass ratio is. We do know that an alpha's q/m is about 4.8E7 C/kg, and a beta's is 1.7E11 C/kg. So maybe we could say that since the alpha's q/m is smaller, its radius should be larger (and it is, in the diagram).
However, I don't think this is valid either, since we also know that alpha particles travel more slowly than betas in general. You could only say that q/m is responsible for the radius if speed was constant, and it isn't.
What am I missing here?
- When you are answering this, suppose you know that , in general, alpha particles are less energetic than beta particles. However, you don't know how many times less energetic the alpha is.
- Assume you know the mass and charge of betas and alphas.
Momentum
p = qBr
As long as q and B are constant, the greater r is, the greater p will be. However, q is not constant here, since alphas carry double the charge magnitude of betas. So I'm not sure you can confidently say that momentum is the reason the alpha and beta have different radii. Am I wrong here?
Charge to Mass Ratio
q/m = v/(Br)
As long as v and B are constant, the greater r is, the smaller the charge-to-mass ratio is. We do know that an alpha's q/m is about 4.8E7 C/kg, and a beta's is 1.7E11 C/kg. So maybe we could say that since the alpha's q/m is smaller, its radius should be larger (and it is, in the diagram).
However, I don't think this is valid either, since we also know that alpha particles travel more slowly than betas in general. You could only say that q/m is responsible for the radius if speed was constant, and it isn't.
What am I missing here?