Discussion Overview
The discussion revolves around the effectiveness of titration as a method for determining the concentrations of acids and bases, specifically focusing on the titration of a weak acid (acetic acid, HAc) with a strong base (sodium hydroxide, NaOH) and the challenges associated with titrating weak acids with weak bases. Participants explore the implications of equilibrium constants and the behavior of pH during titration.
Discussion Character
- Exploratory
- Technical explanation
- Debate/contested
- Mathematical reasoning
Main Points Raised
- One participant questions whether the reaction between NaOH and HAc has an equilibrium constant that is effectively infinite, suggesting that this is necessary for accurate titration results.
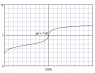
- Another participant clarifies that while the equilibrium constant for the reaction is not infinite, it is likely very large, which allows for a significant pH change at the equivalence point.
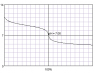
- Concerns are raised about the ability to determine the concentration of a weak acid using a weak base, with participants noting that the equilibrium constant for such reactions is not very large, leading to potential errors in titration results.
- Some participants discuss the importance of the pKa values of the acids and bases involved, noting that if they are close together, it may be difficult to find a suitable indicator for the titration.
- There is a discussion about the mathematical relationships governing the titration process, including the use of formulas relating the normalities and volumes of the acid and base.
- One participant suggests that the overall equilibrium constant for the neutralization reaction of a weak acid with a weak base is not large enough to ensure complete reaction without significant excess of one reactant.
Areas of Agreement / Disagreement
Participants express differing views on the nature of equilibrium constants in acid-base reactions, particularly regarding the effectiveness of titration with weak acids and bases. There is no consensus on the implications of these constants for titration accuracy.
Contextual Notes
Participants note that the assumptions regarding equilibrium constants and the behavior of indicators during titration may depend on specific conditions and definitions, which remain unresolved in the discussion.