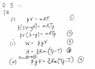Homework Help Overview
The discussion revolves around a thermodynamics problem involving two cylinders and the transfer of gas between them. The original poster explores the relationships between internal energy, work, and heat transfer for a monoatomic ideal gas, questioning the assumptions made regarding the process type and the equations used.
Discussion Character
- Conceptual clarification, Assumption checking, Mathematical reasoning
Approaches and Questions Raised
- The original poster attempts to reconcile the equations for internal energy and work, expressing confusion over the implications of certain assumptions, particularly regarding adiabatic versus isobaric processes.
- Some participants question the validity of equating internal energy and work, suggesting that this relationship holds only under specific conditions.
- Others raise concerns about the definitions and conditions of the process, particularly the pressure dynamics and equilibrium states of the gas in the cylinders.
Discussion Status
The discussion is ongoing, with participants providing insights and clarifications regarding the thermodynamic equations and the nature of the process. There is a recognition of differing interpretations of the problem setup, particularly concerning whether heat transfer can be ignored.
Contextual Notes
Participants note that the problem specifies an isobaric process, yet there are indications of non-equilibrium conditions during the gas transfer. The original poster's confusion stems from the apparent contradiction in the equations and the assumptions about heat transfer.