Discussion Overview
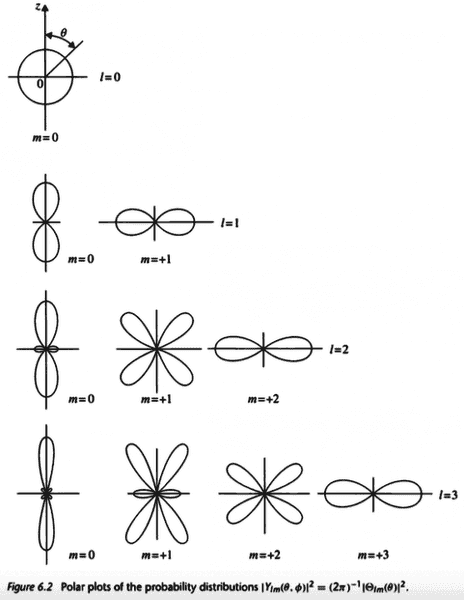
The discussion revolves around the shapes of electron orbitals, specifically whether they are always spherical or if they can take on different geometries, such as the dumbbell shape of the 2p_x orbital. Participants explore the implications of electron excitation, the role of coordinate systems, and the influence of external fields on orbital shapes and spin states.
Discussion Character
- Debate/contested
- Conceptual clarification
- Technical explanation
Main Points Raised
- Some participants question the characterization of the 2p_x orbital as having a dumbbell shape, suggesting that the z-axis can point in any direction, which complicates the interpretation of orbital shapes.
- There is a discussion about the nature of spherical harmonics and their relationship to the 2p_x orbital, with some asserting that the 2p_x is a real function and not an eigenfunction of the angular momentum operator.
- One participant mentions that while orbitals may appear spherical on average, specific conditions, such as polarization of the sample, can lead to non-spherical distributions.
- There is an inquiry into the nature of electron spin, with some asserting that it exists in a superposition of states rather than having a definite direction, and questioning how such superpositions can be prepared.
- Another participant states that there is always a coordinate system in which the spin can be aligned along the z-axis, but this may change in the presence of external fields due to spin precession.
- One participant adds that the choice of which orbital gets excited is influenced by the polarization of the electromagnetic field used for excitation, indicating that the quantization axes can be chosen based on the experimental setup.
Areas of Agreement / Disagreement
Participants express differing views on the shapes of orbitals and the nature of electron spin. There is no consensus on whether orbitals can be considered spherical under all conditions, and the discussion remains unresolved regarding the implications of external fields and the preparation of superpositions.
Contextual Notes
Participants note that the interpretation of orbital shapes may depend on the specific experimental conditions and the definitions used, highlighting the complexity of the topic.