cesiumfrog
- 2,010
- 5
Why is it that neutrons evaporate from nuclei more easily than protons do?
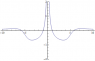
Intuitively, since protons are electrostatically repelled (in addition to whatever nuclear forces they have in common with neutrons), one would expect protons to be ejected more readily than neutrons. (Maybe this is even what does happen for small nuclei, but apparently not large nuclei.)
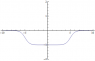
It seems to be said in common parlance that the Coulomb force/barrier acts to contain the protons. Which is counter-intuitive.
On the other hand, at least some textbooks acknowledge the coulomb force trying to push protons away from others in the nucleus, and so they infer that the nuclear force (e.g., residual strong force) must act more strongly on protons than what it does on neutrons. (At least in big nuclei, "beta stable nuclei", nuclei with an excess of neutrons..) So how does this nuclear force distinguish between protons and neutrons?
Intuitively, since protons are electrostatically repelled (in addition to whatever nuclear forces they have in common with neutrons), one would expect protons to be ejected more readily than neutrons. (Maybe this is even what does happen for small nuclei, but apparently not large nuclei.)
It seems to be said in common parlance that the Coulomb force/barrier acts to contain the protons. Which is counter-intuitive.
On the other hand, at least some textbooks acknowledge the coulomb force trying to push protons away from others in the nucleus, and so they infer that the nuclear force (e.g., residual strong force) must act more strongly on protons than what it does on neutrons. (At least in big nuclei, "beta stable nuclei", nuclei with an excess of neutrons..) So how does this nuclear force distinguish between protons and neutrons?