aname
- 8
- 0
- Homework Statement
- i'm not sure I used the numbers right
- Relevant Equations
- Determination of the dissolution enthalpy
-(7.455 so,94+8,4) Delta T

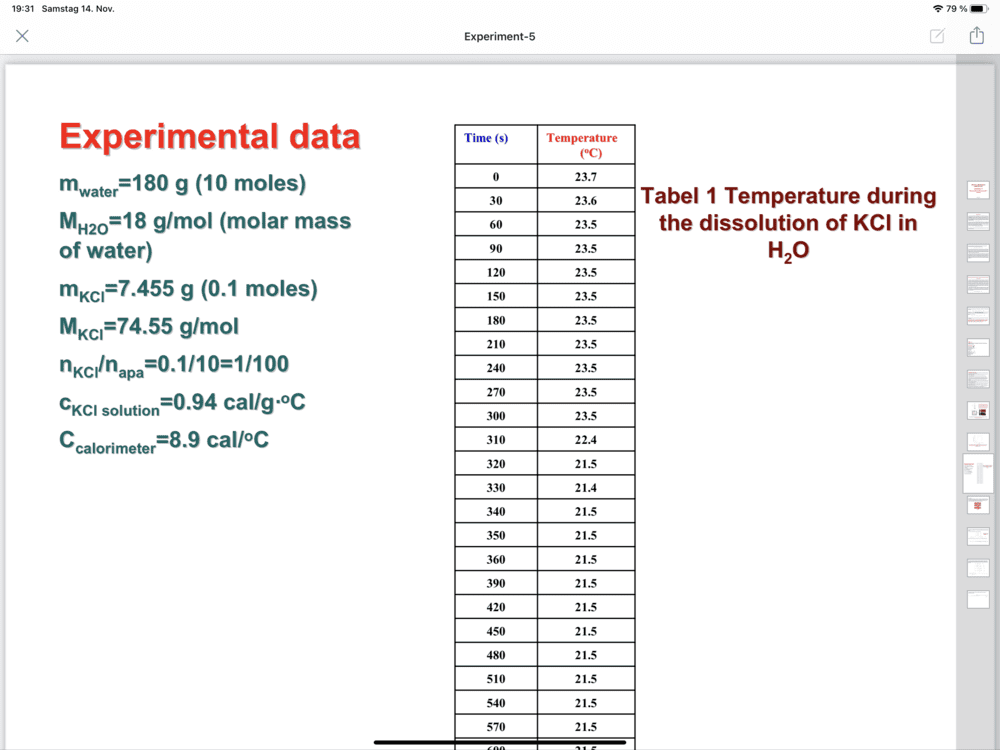
The discussion revolves around the calculation of ΔH (enthalpy change) using a specific formula. Participants are attempting to clarify the components of the formula and the values to be used, including mass and specific heat capacities.
Participants do not appear to reach consensus on the initial calculation or the values used, with multiple viewpoints on the mass of the solution and the formula's application remaining contested.
There are unresolved questions regarding the definitions of terms and the assumptions behind the values used in the calculation, particularly concerning the specific heat capacities and their relevance to the overall formula.
Hi,Chestermiller said:No. m of the solution is 187.5 g.