Discussion Overview
The discussion centers around the calculations involved in nuclear reactions, specifically the merging of isotopes such as deuterium. Participants explore how to mathematically determine the products of such reactions and delve into related concepts like the temperature of isotopes and the Coulomb barrier.
Discussion Character
- Exploratory
- Technical explanation
- Debate/contested
Main Points Raised
- One participant seeks guidance on calculating the products of nuclear reactions involving isotopes like deuterium, expressing a desire for mathematical explanations.
- Another participant provides a link to a Wikipedia article on nuclear reactions, suggesting it as a resource for understanding the topic.
- A participant expresses confusion about calculating the temperature of individual isotopes, questioning the relevance of their inquiry to the original topic.
- Some participants clarify that isotopes do not possess temperatures as individual entities, emphasizing that temperature is a property of a collection of particles.
- One participant introduces the concept of the Coulomb barrier, discussing its role in nuclear reactions and how it relates to the energy required for nuclei to merge.
- Another participant explains that the Coulomb barrier is not a temperature but rather an electric potential energy that affects the likelihood of nuclear reactions occurring.
Areas of Agreement / Disagreement
Participants express differing views on the concept of temperature in relation to isotopes, with some asserting that isotopes do not have temperatures while others attempt to reconcile this with the concept of the Coulomb barrier. The discussion remains unresolved regarding the calculation of temperatures and the implications of the Coulomb barrier in nuclear reactions.
Contextual Notes
There are limitations in the discussion regarding the assumptions made about temperature and its relation to isotopes, as well as the mathematical steps involved in calculating nuclear reactions and the Coulomb barrier.
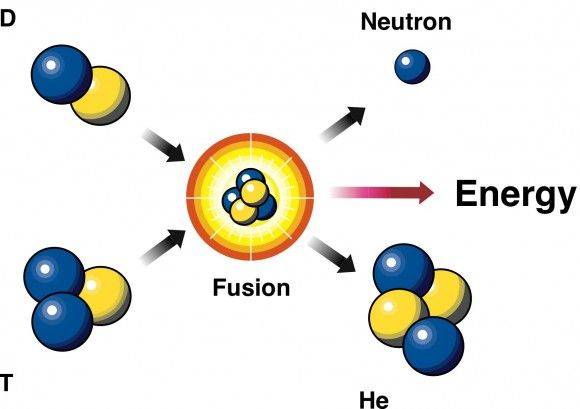
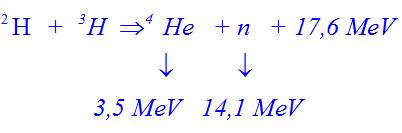 Can someone explain me the calculations? I'm new to the forum, please!
Can someone explain me the calculations? I'm new to the forum, please!