Discussion Overview
The discussion revolves around the final state of a concentration cell, exploring the conditions under which equilibrium is reached, the relationship between voltage and concentration, and the implications of connecting multiple vats with different concentrations. Participants examine both theoretical and practical aspects of concentration cells and their behavior in various configurations.
Discussion Character
- Exploratory
- Technical explanation
- Conceptual clarification
- Debate/contested
- Mathematical reasoning
Main Points Raised
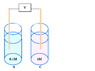
- Some participants propose that the final state of a concentration cell is reached when the concentrations are equal, while others clarify that it is technically when the activities are the same, which may involve different concentrations due to ionic atmospheres.
- One participant discusses the relationship between current and voltage, suggesting that current is linearly related to voltage under constant load, and presents a mathematical model using the Nernst equation.
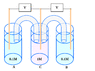
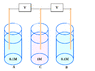
- A later reply questions how the voltage behaves in a system with multiple vats of different concentrations and whether the voltage measured across voltmeters connected to these vats would be a fraction of the equilibrium voltage of a simpler concentration cell.
- There is a debate about the chemical processes occurring in each compartment, with some participants asserting that compartment C undergoes reduction while A and B undergo oxidation.
- One participant raises a concern about the effects of removing salt bridges, suggesting that this could lead to an electroneutrality violation that halts chemical reactions and affects voltage measurements.
Areas of Agreement / Disagreement
Participants express differing views on the final state of concentration cells, the implications of connecting multiple vats, and the chemical processes involved. The discussion remains unresolved with multiple competing perspectives and no consensus reached.
Contextual Notes
Participants note the importance of ionic atmospheres and the potential for different activities affecting the final state. There are also mentions of assumptions regarding the behavior of voltages and currents in the described configurations.
Who May Find This Useful
This discussion may be of interest to those studying electrochemistry, concentration cells, and the behavior of voltages in multi-compartment systems, as well as individuals exploring the theoretical underpinnings of these concepts.