Neolux
- 5
- 0
I am making a Geometric model for VESPR theory, which states that valence electron pairs are mutually repulsive, and therefore adopt a position which minimizes this, which is the position at which they are farthest apart, still in their orbitals.
For example, the 2 electron pairs on either side of the central atom of Carbon Dioxide both repel each other equally (Dot Diagram Below), giving the molecule a linear arrangement (Shown Below):
Dot Diagram of Carbon Dioxide:
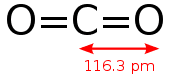
Linear Arrangement:

Other molecules like water molecules have 4 pairs (2 bonded pairs, 2 unbonded pairs), which all repel each other, giving it a bent configuration.
Dot Diagram of Water:

Bent Arrangement:
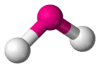
I figured it'd be pretty simple and straightforward to create a geometric interpretation of this. This has probably already been done, but just doing it for fun.
I figured I could accomplish this by creating a 3-D space, plotting the pairs as points, and then using the 3-D distance formula, find the arrangement at which all the pairs are at the greatest distance (Equal distance) from each other. I could then also calculate the angle of the bonds by calculating the angle between those lines.
All the points would have to be an equal distance from the nucleus (origin), which can be set to an arbitrary distance. Let's just say 1.
Here is what I have so far (For a simple 2 pair configuration):
__________________________________________________________________________________
E1 = Electron Pair 1
E2 = Electron Pair 2
Origin = Nucleus
Distance from E1 to Nucleus = Distance from E2 to Nucleus:
√(E1x-0)^2 + (E1y-0)^2 + (E1z-0)^2 = √(E2x-0)^2 + (E2y-0)^2 + (E2z-0)^2
Distance from E1 to E2 (Needs to be maximized):
√(E1x-E2x)^2 + (E1y-E2y)^2 + (E1z-E2z)^2
__________________________________________________________________________________
Now how could I calculate the coordinates of E1x, E1y, E1z, E2x, E2y, and E2z such that the distance between them is a maximum, yet they are the same distance from the origin? I figured i'd need differential geometry for that bit.
Once I find the coordinates for each pair, I figured I could then find the angle between them using this:
__________________________________________________________________________________
Θ = tan^-1(m1-m2/1+m1m2)
__________________________________________________________________________________
Where m1 and m2 are the slopes of the lines from each point to the origin.
Any help or comments would be greatly appreciated.
Thanks in advance.
For example, the 2 electron pairs on either side of the central atom of Carbon Dioxide both repel each other equally (Dot Diagram Below), giving the molecule a linear arrangement (Shown Below):
Dot Diagram of Carbon Dioxide:
Linear Arrangement:
Other molecules like water molecules have 4 pairs (2 bonded pairs, 2 unbonded pairs), which all repel each other, giving it a bent configuration.
Dot Diagram of Water:
Bent Arrangement:
I figured it'd be pretty simple and straightforward to create a geometric interpretation of this. This has probably already been done, but just doing it for fun.
I figured I could accomplish this by creating a 3-D space, plotting the pairs as points, and then using the 3-D distance formula, find the arrangement at which all the pairs are at the greatest distance (Equal distance) from each other. I could then also calculate the angle of the bonds by calculating the angle between those lines.
All the points would have to be an equal distance from the nucleus (origin), which can be set to an arbitrary distance. Let's just say 1.
Here is what I have so far (For a simple 2 pair configuration):
__________________________________________________________________________________
E1 = Electron Pair 1
E2 = Electron Pair 2
Origin = Nucleus
Distance from E1 to Nucleus = Distance from E2 to Nucleus:
√(E1x-0)^2 + (E1y-0)^2 + (E1z-0)^2 = √(E2x-0)^2 + (E2y-0)^2 + (E2z-0)^2
Distance from E1 to E2 (Needs to be maximized):
√(E1x-E2x)^2 + (E1y-E2y)^2 + (E1z-E2z)^2
__________________________________________________________________________________
Now how could I calculate the coordinates of E1x, E1y, E1z, E2x, E2y, and E2z such that the distance between them is a maximum, yet they are the same distance from the origin? I figured i'd need differential geometry for that bit.
Once I find the coordinates for each pair, I figured I could then find the angle between them using this:
__________________________________________________________________________________
Θ = tan^-1(m1-m2/1+m1m2)
__________________________________________________________________________________
Where m1 and m2 are the slopes of the lines from each point to the origin.
Any help or comments would be greatly appreciated.
Thanks in advance.