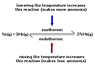
SUMMARY
Increasing the temperature of an endothermic reaction results in a higher yield of products due to Le Chatelier's principle, which states that the system will shift to absorb the added energy. Conversely, decreasing the temperature favors exothermic reactions, leading to an increase in product formation as the system releases energy to counteract the temperature drop. This understanding is crucial for manipulating reaction conditions in chemical processes.
PREREQUISITES
- Le Chatelier's principle
- Endothermic and exothermic reactions
- Activation energy concepts
- Basic thermodynamics
NEXT STEPS
- Study the implications of Le Chatelier's principle in chemical equilibrium
- Explore the effects of temperature changes on reaction kinetics
- Investigate specific endothermic and exothermic reactions
- Learn about activation energy and its role in reaction rates
USEFUL FOR
Chemistry students, chemical engineers, and anyone interested in understanding reaction dynamics and equilibrium principles.