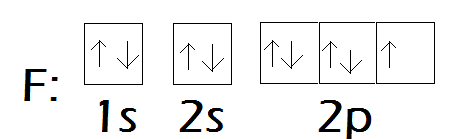Discussion Overview
The discussion revolves around the concept of back-bonding in phosphorus trifluoride (PF3), exploring the hybridization of phosphorus and fluorine, and the implications for bonding interactions. Participants examine theoretical frameworks, computational analyses, and differing views on the nature of bonding in PF3.
Discussion Character
- Exploratory
- Technical explanation
- Debate/contested
- Mathematical reasoning
Main Points Raised
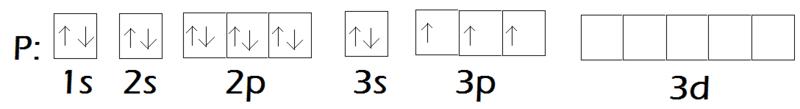
- Some participants assert that phosphorus is sp3 hybridized and fluorine is also sp3 hybridized, suggesting that back-bonding can only occur with pure, unhybridized orbitals.
- Others question the hybridization of fluorine, expressing uncertainty about its classification.
- A participant argues that back-bonding is likely in PF3 due to phosphorus having vacant 3d orbitals, despite its sp3 hybridization, and that fluorine has lone pairs available for bonding.
- Another participant contends that hybridization is a conceptual tool rather than a strict property of atoms, suggesting that the energetic differences in fluorine's orbitals make hybridization unnecessary.
- A computational analysis using Gaussian 09W and NBO indicates that PF3 has a high Lewis structure occupancy, with detailed percentages of orbital contributions from phosphorus and fluorine, suggesting a more complex hybridization than simple sp3.
- Some participants clarify that small contributions from d-orbitals do not necessarily indicate back-bonding but may serve as polarization functions, emphasizing the distinction between density functional theory (DFT) and valence bond theory (VB).
- There is a mention of the complexity involved in analyzing back-bonding through NBO calculations, with a suggestion that certain interactions need to be turned off to accurately assess back-bonding energy.
Areas of Agreement / Disagreement
Participants express differing views on the hybridization of fluorine and the nature of back-bonding in PF3. There is no consensus on the role of d-orbitals or the validity of hybridization as a concept in this context, indicating ongoing debate and uncertainty.
Contextual Notes
Limitations include the dependence on definitions of hybridization, the unresolved nature of back-bonding analysis, and the distinction between computational methods (DFT vs. VB) that may affect interpretations of bonding interactions.