How many Rb atoms to fit on a 9Cm^2 square?
Click For Summary
Discussion Overview
The discussion revolves around calculating the number of rubidium (Rb) atoms that can fit on a 9 cm² square. Participants explore various mathematical approaches and geometric considerations related to atomic spacing and arrangement.
Discussion Character
- Exploratory
- Mathematical reasoning
- Homework-related
Main Points Raised
- One participant suggests calculating the area of a single Rb atom using its radius and dividing the total area by this value to estimate the number of atoms, questioning the validity of a previous estimate of 4.7 x 10^15 atoms.
- Another participant points out that empty spaces between atoms must be considered, indicating that the simple area calculation is insufficient.
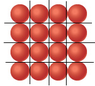
- There is a discussion about the geometric arrangement of atoms, with suggestions to use equilateral triangles or parallelograms to model the atomic structure, though participants express uncertainty about the calculations involved.
- Some participants seek clarification on how to draw diagrams to visualize the problem, indicating a need for visual aids in understanding the spatial arrangement of atoms.
- A participant expresses frustration over the complexity of the problem and requests stronger hints to aid in their understanding.
- One participant claims to have solved the problem, though the details of their solution are not shared in the discussion.
Areas of Agreement / Disagreement
Participants do not reach a consensus on the best method to calculate the number of Rb atoms fitting in the given area, with multiple competing views on the geometric arrangements and calculations involved.
Contextual Notes
Participants mention various geometric shapes and arrangements (triangles, parallelograms) but do not resolve the mathematical steps necessary to finalize their calculations. There is also uncertainty regarding the assumptions about atomic spacing.
Similar threads
- · Replies 1 ·
- · Replies 3 ·
Graduate
Probability of resonant ionization
- · Replies 18 ·
- · Replies 1 ·
- · Replies 2 ·
- · Replies 1 ·
- · Replies 5 ·