Discussion Overview
The discussion centers around the utility of Molecular Orbital Theory in understanding molecular structures and bond orders, particularly in complex molecules like BF3 with a higher electron count. Participants explore the relevance of orbital hybridization and geometry in this context.
Discussion Character
- Exploratory
- Technical explanation
- Conceptual clarification
- Homework-related
Main Points Raised
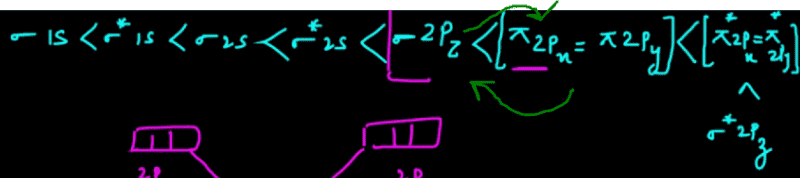
- One participant expresses confidence in the usefulness of Molecular Orbital Theory but notes a limitation in available orbital information, specifically mentioning the σ*2pz orbital.
- Another participant suggests that understanding the geometry and symmetry of molecules is important, referencing sp3 hybridization and its implications for molecular shape and lone pairs.
- A participant reiterates their initial question about determining bond order in BF3 and seeks guidance on how to approach the problem.
- Links to external resources are provided by participants as potential starting points for further exploration of the topic.
- One participant acknowledges the complexity of the provided resources, indicating a struggle to grasp the material.
Areas of Agreement / Disagreement
Participants do not reach a consensus on the effectiveness of Molecular Orbital Theory or the best methods for calculating bond order in complex molecules. Multiple viewpoints and levels of understanding are present throughout the discussion.
Contextual Notes
Some participants express uncertainty regarding the application of Molecular Orbital Theory to specific molecules and the complexity of the concepts involved. There is also a reliance on external resources that may not fully address the participants' questions.
Who May Find This Useful
This discussion may be of interest to students and educators in chemistry and physics, particularly those exploring molecular structures, orbital theory, and hybridization concepts.