Discussion Overview
The discussion revolves around the interpretations of diagrams representing the molecular structures of Carbon Monoxide (CO) and Carbon Dioxide (CO2), focusing on hybridization, bonding, and the accuracy of the diagrams presented. Participants are seeking to clarify their understanding of these concepts.
Discussion Character
- Exploratory
- Technical explanation
- Debate/contested
- Mathematical reasoning
Main Points Raised
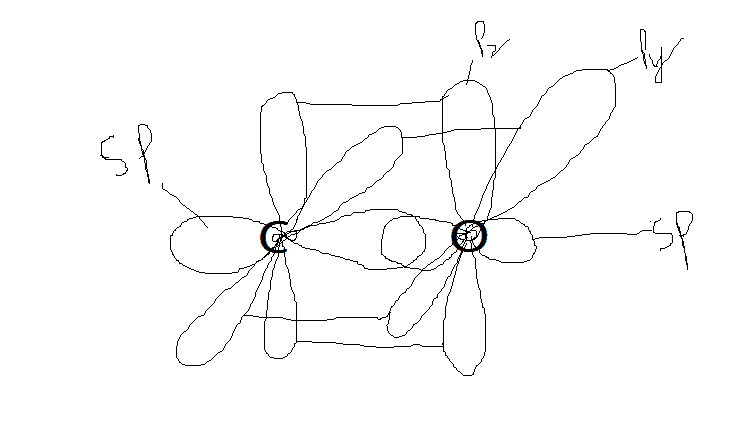
- One participant questions the accuracy of their diagrams for CO and CO2, specifically regarding hybridized orbitals and bonding representations.
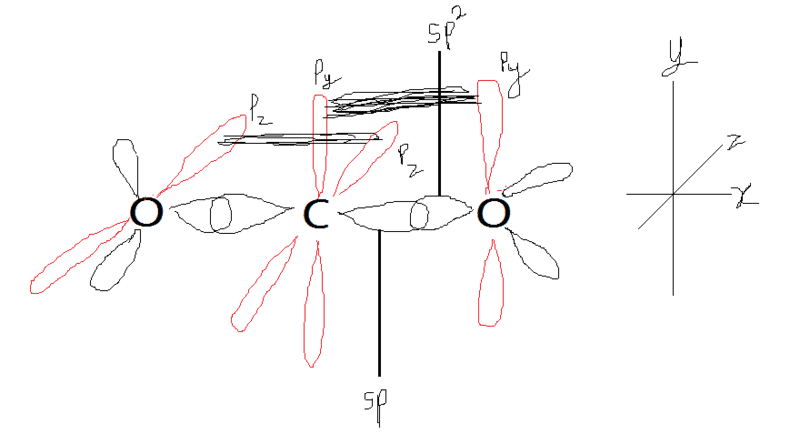
- Another participant suggests that while the orbitals are mostly correct, there are details that need improvement, particularly in the representation of bonding for CO2.
- A participant mentions that the bond order in CO cannot be clearly represented in the diagram due to its resonance structure.
- There is a discussion about whether side atoms or ligands, such as oxygen in CO, exhibit hybridization, with some participants asserting that they do not.
- One participant introduces the idea that different assumptions about hybridization can lead to various bonding schemes for CO2, presenting multiple models including sp2 and sp hybridization for oxygen.
- Another participant discusses the complexity of hybridization in larger atoms and mentions hypervalency in compounds like PF5, contrasting it with the simpler bonding in CO and CO2.
- Alternative bonding models, such as considering oxygens as unhybridized or sp3 hybridized, are also proposed, along with the concept of "banana bonds" and their implications for bonding strength.
Areas of Agreement / Disagreement
Participants express differing views on the hybridization of ligands and the representation of bonding in CO and CO2. Multiple competing models regarding hybridization and bonding schemes are presented, and the discussion remains unresolved.
Contextual Notes
Some participants note limitations in the diagrams, such as missing color coding for hybridized and unhybridized orbitals, and the complexity of hybridization in larger atoms compared to those in the second period.