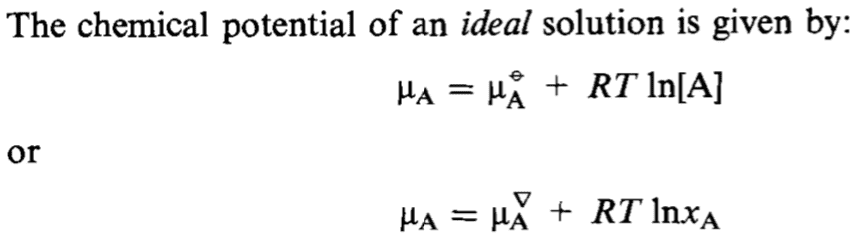Discussion Overview
The discussion revolves around the meaning of a small triangle symbol in the context of chemical potential, particularly in relation to ideal solutions. Participants explore its significance in different definitions and notations used in chemistry, referencing both theoretical and practical aspects of chemical potential.
Discussion Character
- Exploratory
- Technical explanation
- Debate/contested
Main Points Raised
- Some participants note that the small triangle symbol is unfamiliar and may represent outdated or obscure notation.
- One participant suggests that the triangle could indicate a reference state, contrasting a solution with a concentration of [A] = 1M and a pure solvent state where xA = 1.
- A later reply proposes that the symbol might denote the chemical potential of a pure solvent A, indicating a difference from the IUPAC recommended notation.
- Another participant provides a detailed explanation of the chemical potential equations, relating them to vapor pressure and activity coefficients, and connects these to the original question about the small triangle.
Areas of Agreement / Disagreement
Participants express uncertainty regarding the meaning of the small triangle symbol, and no consensus is reached on its significance or whether it is a standard notation.
Contextual Notes
There are references to different notations and definitions that may not align with IUPAC recommendations, indicating potential limitations in understanding or interpreting the symbol's meaning.