Abhi9826
- 7
- 1
I just wonder that why the negative deviation comes in graph except for hydrogen and helium gas.I am still thinking for the reason and searching for it.So please help me ...
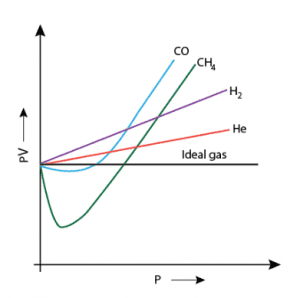
The discussion revolves around the negative deviation observed in the PV vs P graph for various gases, particularly focusing on why hydrogen and helium do not exhibit this behavior. Participants explore theoretical explanations, mathematical models, and the implications of gas liquefaction on the observed phenomena.
Participants express differing views on the reasons behind the negative deviation in the graph, with some attributing it to liquefaction and others focusing on mathematical modeling. No consensus is reached regarding the exact causes or the validity of the graph's representation.
Limitations include the lack of specific temperature data for the graph and the dependence on the assumptions made in the mathematical models discussed. The discussion also highlights the complexity of gas behavior under varying conditions.
An interesting interpretation. Without additional information, such as the temperature at which the data in the graph are supposed to represent, it is difficult to ascertain exactly what properties the graph is trying to illustrate.John Park said:To provide a different sort of answer: the deviations from linearity are to do with the gases liquifiying. Hydrogen and helium don't become liquids until the temperature is very low, and may not show that behaviour at the temperatures represented in the plot.
The product PV is closely related to the so-called compressibility factor z = PV/nT. Its behaviour was (is?) an important topic in studies of the macroscopic properties of fluids.
In fact if volumes, temperatures, and pressures are scaled to the corresponding "critical" values (i.e. values for the state where liquid and gas are indistinguishable), most gases show very similar compressibility factors, over reasonable ranges of pressure. Look up the Law (or Theorem) of Corresponding States for more details.
Edit: Wikipedia has a good article on compressibility factors. The behaviours of hydrogen and helium are influenced by quantum effects.