Homework Help Overview
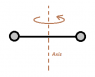
The discussion revolves around the moment of inertia of an oxygen molecule, specifically estimating the effective distance between its two atoms based on given mass and moment of inertia values. The subject area includes concepts from rotational dynamics and geometry.
Discussion Character
- Exploratory, Conceptual clarification, Mathematical reasoning, Problem interpretation, Assumption checking
Approaches and Questions Raised
- Participants explore the relationship between the calculated radius and the actual distance between the atoms, questioning the geometric interpretation of the radius. There are attempts to clarify the definitions of moment of inertia and moment arm, as well as to relate them to the problem at hand.
Discussion Status
Participants are actively engaging with the problem, raising questions about the definitions and relationships involved. Some have identified potential misunderstandings regarding the use of moment of inertia in geometric equations. There is a recognition of the need to clarify the roles of different variables in the context of the problem.
Contextual Notes
There appears to be confusion regarding the application of trigonometric relationships and the distinction between moment of inertia and moment arm, which may affect the interpretation of the problem. Participants are working within the constraints of the information provided and the definitions of physical quantities involved.