Homework Help Overview
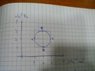
The discussion revolves around a thermodynamics problem involving a monoatomic ideal gas undergoing a quasi-static p-v cycle represented on a p-v diagram. The cycle includes states A, B, C, and D, with specific pressures and volumes given. Participants are tasked with determining the work done by the gas throughout the cycle.
Discussion Character
- Exploratory, Conceptual clarification, Mathematical reasoning, Problem interpretation, Assumption checking
Approaches and Questions Raised
- Participants discuss the nature of the processes between states, questioning the classification of certain segments as isothermal or adiabatic. There are attempts to relate the area under the curve on the p-v diagram to the work done by the gas. Some participants express confusion about how to calculate work for varying conditions and the implications of temperature changes on internal energy.
Discussion Status
Guidance has been offered regarding the relationship between the area under the curve and work done, as well as the need to consider the specific paths taken in the cycle. Multiple interpretations of the problem are being explored, particularly concerning the calculations for work and heat transfer along different segments of the cycle.
Contextual Notes
Participants note potential confusion regarding terminology, such as "liquid heat," and the implications of the symmetry of the cycle on calculations. There is also mention of the challenge posed by varying temperature and pressure along certain paths, which complicates the integration process for work and heat calculations.