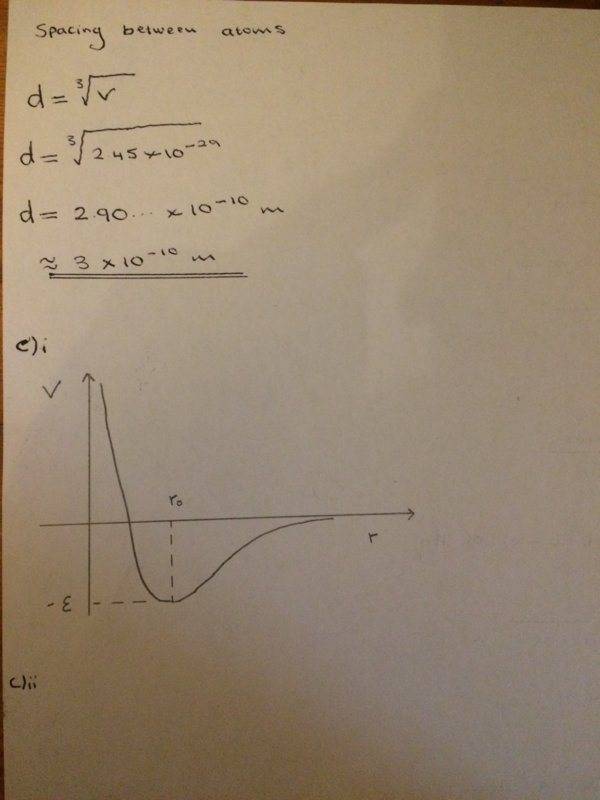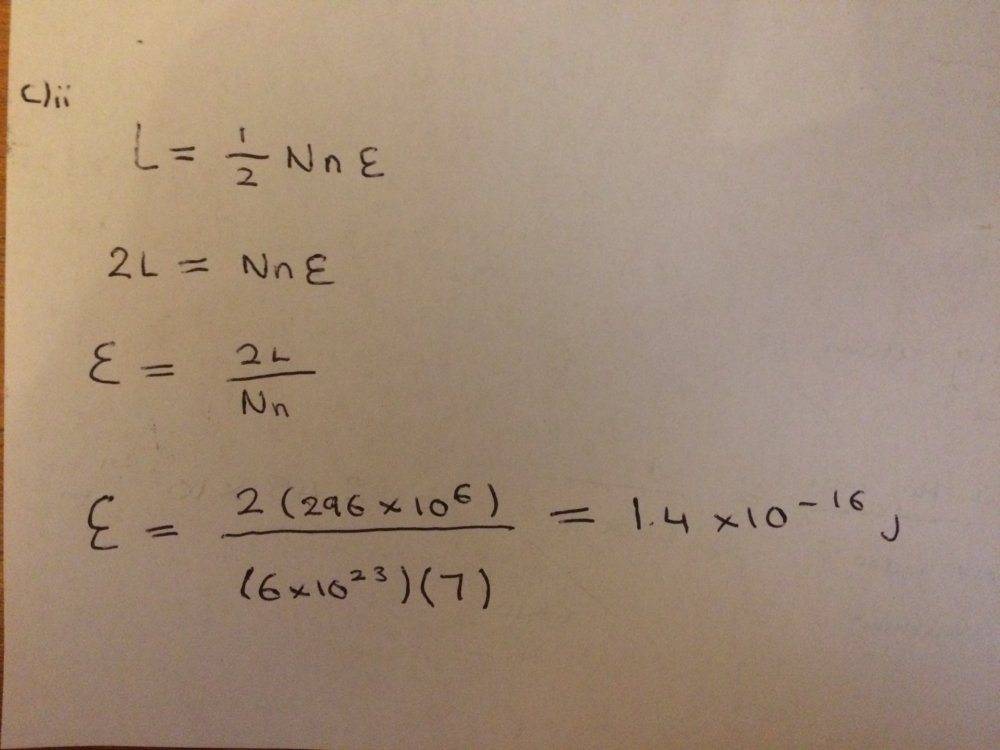Homework Help Overview
The discussion revolves around calculating binding energy and related concepts in a chemistry context, specifically focusing on part c)ii of a homework problem. Participants are exploring unit conversions and calculations involving energy and volume per atom.
Discussion Character
Approaches and Questions Raised
- Participants discuss the conversion of energy units from kJ/kg to kJ/mol and the implications for calculating binding energy. There are attempts to clarify calculations and unit tracking, alongside questions about the approach to part d)ii.
Discussion Status
Some participants have provided guidance on unit conversions and calculation methods, while others are exploring different interpretations of the problem. There is acknowledgment of potential confusion regarding the substances involved in the calculations.
Contextual Notes
Participants note the importance of unit consistency in calculations and the need to clarify which substance is being discussed, as there is a mix-up between mercury and neon. There is also mention of specific values and constants that are relevant to the calculations.