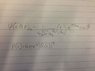SUMMARY
The discussion focuses on calculating the expectation value for a hydrogen atom in the quantum state defined by n=2, l=1, and m=0. The wave function is given as ψ(r,θ,∅)=(1/4(√2pi)ab3/2)(r/ab)(e-r/2ab)(cos(θ). To find , participants emphasize the need to derive the radial function R(r) from the wave function and subsequently use it to compute the probability density P(r) = 4πr²|R(r)|². The integral for is established as = ∫₀^∞ P(r) r³ dr, incorporating the necessary factors for spherical coordinates.
PREREQUISITES
- Understanding of quantum mechanics, specifically hydrogen atom wave functions.
- Familiarity with spherical coordinates in three-dimensional integrals.
- Knowledge of probability density functions in quantum mechanics.
- Ability to perform integrals involving spherical harmonics and radial functions.
NEXT STEPS
- Study the derivation of radial wave functions for hydrogen-like atoms.
- Learn about the normalization of wave functions in quantum mechanics.
- Explore the application of spherical harmonics in quantum state calculations.
- Investigate the significance of expectation values in quantum mechanics.
USEFUL FOR
Students and educators in quantum mechanics, physicists working with atomic models, and anyone interested in the mathematical foundations of wave functions and expectation values in quantum systems.