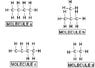
Which Structure Has The Highest Melting Point?
- Thread starter ProblemSets
- Start date
Click For Summary
Discussion Overview
The discussion revolves around identifying which molecular structure has the highest melting point, exploring the molecular features that influence melting points, and examining the types of intermolecular forces involved.
Discussion Character
- Exploratory, Technical explanation, Debate/contested, Homework-related
Main Points Raised
- One participant asks which structure has the highest melting point.
- Another participant inquires about the molecular features that determine melting points.
- Several participants mention different intermolecular forces, including hydrogen bonding, dipole-dipole interactions, and London dispersion forces.
- One participant suggests that structure C might be correct due to its polar characteristics and longer carbon chain.
- Another participant challenges the correctness of structure C, indicating it is not the right answer.
- A participant expresses uncertainty and suggests that structure A may be the correct answer after conducting research.
- Another participant questions the research methods used to arrive at the conclusion.
Areas of Agreement / Disagreement
Participants do not reach a consensus on which structure has the highest melting point, with multiple competing views and corrections being presented throughout the discussion.
Contextual Notes
Participants reference different types of intermolecular forces and their relative strengths, but the discussion does not resolve which specific structure is correct or the assumptions behind their claims.
Who May Find This Useful
This discussion may be useful for students or individuals interested in chemistry, particularly in understanding the factors affecting melting points and the nature of intermolecular forces.
Similar threads
- · Replies 3 ·
- · Replies 5 ·
- · Replies 15 ·
- · Replies 9 ·
- · Replies 5 ·
- · Replies 5 ·
- · Replies 1 ·