Discussion Overview
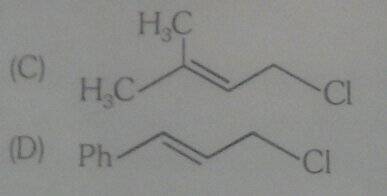
The discussion centers around the rates of solvolysis of two compounds, C and D, in a 50% aqueous ethanol solution at 45°C. Participants explore the factors influencing the rates, including resonance effects and the nature of the solvent.
Discussion Character
- Debate/contested
- Technical explanation
- Conceptual clarification
Main Points Raised
- One participant claims that compound D should have a higher rate of solvolysis due to more resonance, yet observes that compound C exhibits a higher rate.
- Several participants seek clarification on the definition of the solvent in the context of solvolysis, emphasizing that solvolysis involves a reaction with the solvent.
- A participant clarifies that the solvent is a 50% ethanol and water mixture, which is intended to enhance the solubility of the unsaturated halides involved in the reaction.
- Another participant suggests that the phenyl substituted structure may destabilize the carbon-chloride bond more effectively than the other compound, which lacks resonance.
- There is a discussion about the reaction mechanisms, with one participant noting that the reaction may proceed via an Sn2 process due to the presence of a primary carbon, while also referencing their experiences with tertiary halides in alcohol/water mixtures.
- One participant expresses a desire to see experimental results to confirm the theoretical discussions about the rates of solvolysis.
Areas of Agreement / Disagreement
Participants express differing views on which compound exhibits a higher rate of solvolysis, with some supporting the idea that resonance plays a significant role, while others question the definitions and mechanisms involved. The discussion remains unresolved regarding the comparative rates of solvolysis for compounds C and D.
Contextual Notes
There are uncertainties regarding the definitions of solvent and substrate, as well as the specific mechanisms (Sn1 vs. Sn2) that may apply to the compounds in question. The discussion also highlights the complexity of predicting reaction rates based on structural features.